Abstract
In the current study, four different curcumin-NSAID conjugates are synthesized, and their biological screening for rheumatoid arthritis, anti-inflammatory, and anti-ulcer properties follows. One phytochemical with a wide range of therapeutic applications is curcumin. In the sense that it may aid in achieving the goal of incorporating specific desired properties in curcumin, such as enhanced bioavailability, reduction in side effects, and also offering good therapeutic potential in terms of determination of rheumatoid arthritis, anti-inflammatory and anti-ulcer activities of synthesized conjugates using appropriate animal models, the idea of conjugating curcumin with various NSAIDs is somewhat novel. When Curcumin and NSAIDs were tested alone, as well as with the usual medication used for biological evaluation, the intended outcomes were achieved. All of the produced compounds' structural characteristics have been validated using suitable spectrum analysis. Thin Layer Chromatography analysis, recrystallization, and melting point measurement are further supporting techniques used for structural and purity issues.
Keywords
Bioavailability, Anti-Inflammatory, Anti-Ulcer, Rheumatoid Arthritis, etc
Introduction
Isolated from the turmeric rhizomes, curcumin is a yellow polyphenolic chemical molecule. Differentiuloyl methane (Curcuma longa) is its chemical name. [1][2][3] The potent antioxidant and anti-inflammatory qualities of this substance have been linked to its therapeutic effectiveness. [4][5] Curcumin has several pharmacological properties, although it is not yet approved as a medication. Its low oral bioavailability (1% or less) is the cause of this. [6] Curcumin's rapid pre-systemic metabolism, poor solubility, permeability, and water stability have all been related to its low bioavailability. [7] Orange-yellow in color, curcumin is a crystalline powder that is almost completely soluble in water. Recently, curcumin has been investigated for treatment of HIV, AIDS, viral infections, scabies, high cholesterol, inflammation, and chronic anterior uveitis, a visual problem. [8][9] Curcuma longa is a drug with a wizard-like appearance that comes from the traditional Indian medical system. In Ayurveda, it is used to treat illnesses with high Pitta and Kapha. [10] The 70 species in the Curcuma genus include Curcuma longa, Curcuma zanthrorrhiza, Curcuma zedoria, Curcuma angustifolia, Curcuma aromatica, and Curcuma caesia, several of which have important therapeutic properties. [11]. Most often used medicinally are its oil, rhizome, and tubers. The rhizomes have a vivid orange inside and a rough brown outside. Rhizomes have a distinct smell and a potent, bitter flavor. They can be colored, elliptical, or rectangular in shape. [12]. The tropical crop curcuma longa is widely grown in Bangladesh, China, India, Malaysia, Indonesia, Thailand, Cambodia, and the Philippines. Kerala, Tamil Nadu, Orissa, Karnataka, Andhra Pradesh, and Maharashtra are the main Indian states that cultivate turmeric. India produces more than 90% of the total amount produced worldwide. [13][14]
CHEMISTRY OF CURCUMIN
Diferuloyl methane, or curcumin, is a symmetric chemical moiety with the molecular weight of 368.38 and the chemical formula C21H20O6. It has the IUPAC name 1E,6E.1-hydroxy-3-methoxyphenyl-1,7-bis 1,6-heptadiene and 3,5-dione. A seven-carbon linker consisting of an unsaturated diketone moiety connects two aromatic ring systems with o-methoxy phenolic groups. [15][16][17][18][19] The diketo group, which can exist in many conformers based on the surroundings, contains keto-enol tautomerism. [20] The most active ingredient in turmeric is curcumin, which makes up 5%–7% of its active components. The characteristic yellow of turmeric is caused by the curcuminoids, which Vogel first discovered in 1842. Curcuma longa naturally contains three different forms of curcuminoids (Curs): bis-demethoxycurcumin (Bis; 5 %), demethoxycurcumin (Dem; 15 %), and curcumin (Cur; diferuloylmethane, 10%). When Lampe and Milobedeska first reported on curcumin (C21H20O6) in 1910, they found that it has a diferuloylmethane structure. [21]
Figure1: Disease target of curcumin
Poor Bioavailability of Curcumin
Inadequate absorption, a high metabolic rate, and/or quick excretion from the body can contribute to a medication's reduced bioavailability. Studies have demonstrated the strong intrinsic activity of curcumin and its efficacy as a medicinal agent for a range of illnesses. Unfortunately, studies conducted over the past three decades on curcumin's distribution, metabolism, excretion, and absorption have shown that curcumin has a rapid metabolism and low absorption, which significantly reduces its bioavailability. [22] It is common knowledge that curcumin has poor pharmacokinetic profile, poor water solubility, and chemical instability. Curcumin is safe and effective, but even at high concentrations, it has poor absorption in humans, raising questions about its usefulness as a medicine. [23] Due to its low rate of absorption in the small intestine, extensive reductive and conjugative metabolism in the liver, and excretion through the gall bladder, curcumin has a restricted oral bioavailability. The limited bioavailability of curcumin is further compounded by its binding to enterocyte proteins, which might change its structure. [24] A number of strategies, such as adjuvants that stop curcumin metabolism and novel solid and liquid oral administration vehicles, have been tried to address the problem of insufficient absorption of curcumin and its quick excretion from the body. [25] By improving solubility, extending plasma residence time, and enhancing pharmacokinetic profile and cellular absorption, these novel drug delivery techniques may be able to resolve the pharmaceutical issue associated with curcumin administration. [26]
NON STEROIDAL ANTI-INFLAMMATORY DRUGS
One of the most generally advised treatments in the globe is the use of NSAIDs. Based on their chemical structure and selectivity, they are divided into the following groups: anthranilic, propionic, acetic, non-acetylated, acetylated, enolic, and selective COX-2 inhibitors. [27]
NSAIDs' primary medicinal effect is to suppress the cyclooxygenase enzymes (COX-1 and COX-2), which stops specific prostaglandins (PGs) from being produced. Thus, medications ought to reduce inflammation and discomfort in the joints while doing less harm to the gastrointestinal tract than non-selective NSAIDs. However, they are well recognized to cause duodenal and stomach ulcers (DU). Moreover, NSAIDs are associated with a high incidence of gastrointestinal (GI) issues, which cause 5–15% of patients to discontinue using them. [28] The reason why NSAIDs cause gastrointestinal side effects is because they inhibit the formation of prostaglandins, which lowers the GI mucosal barrier and raises the risk of bleeding. [28] Curcumin-NSAID conjugates have the ability to mitigate the side effects of long-term usage of non-steroidal anti-inflammatory drugs (NSAIDs) due to curcumin's inherent gastro-protective qualities. [28]
MATERIALS AND METHODS
ISOLATION AND PURIFICATION OF CURCUMIN
Turmeric rhizome powder was extracted in a soxhlet assembly using ethyl acetate until all coloring matter was removed. The crude extract was then condensed into a semisolid dark tinted substance. One gram of crude extract was mixed with 25 mL of hexane for 12 hours. After that, the solution containing a lump of crude extract was agitated for three hours at 600 rpm with a magnetic stirrer. The bulk separated into small fragments during the churning process and was eventually reduced to powder. The suspended lump powder was separated by centrifugation and dried in an oven at 40°C. The total curcumin content of the resulting crude powder was found. Powdered curcuminoid was melted point measured using the open capillary method. To distinguish between pure and crude curcumin, recrystallization was utilized.
TLC separation of curcuminoids using various solvent systems:
Using TLC, three curcuminoids were found in the acetone extract. The TLC pre-coated silica gel plates (Merk-60 F254, 0.25 mm thick) were developed in a Camag twin trough glass tank, with each plate developing to a height of 10 cm after being impregnated with the mobile phase for an hour.To optimize the composition of the mobile phase, a range of mobile solvents with varying polarities were employed. The development plates were taken out and allowed to dry before the spots were examined under a UV lamp.
Silica gel column chromatography
On a glass column filled with silica gel (60–120 mesh), the acetone extract was chromatographed. After adding 5 grams of curcuminoids and 8 grams of silica gel to a 462 cm column, the mixture was eluted with chloroform, chloroform:methanol, and chloroform:methanol with increasing polarity. Using a developing solvent solution of chloroform:methanol (95:5), all of the generated fragments were run on a 60 F254 silica gel TLC plate and recognized as yellow spots. A rotary evaporator was used to remove the organic solvent, and similar fractions with Rf values were combined. For every curcuminoid sample, the total curcuminoid concentration was measured using UV spectrophotometry at 420 nm.
Purification of each curcuminoids:
Using column chromatography, each curcuminoids was extracted and then dissolved in methanol before being heated. Chloroform should be added after complete dissolution to achieve a 5:2 methanol to chloroform ratio. Keep for overnight at 5°C. The crystals were separated via filtering. The crystals were precipitated using petroleum ether. HPLC was used to measure the purity of each individual crystal.
Synthesis of Curcumin-NSAIDs Conjugate:
Aqueous solutions of curcumin (5 g, 13.6 mmol) in dichloromethane:ethyl ether (70:30, 500 ml) were mixed with an equimolar amount of 4-Dimethylaminopyridine (DMAP) and stirred for 30 minutes. The previously described combination was supplemented with various NSAIDs and 1-Ethyl-3-(3-dimethyl aminopropyl) carbodiimide (EDAC) (20.4 mmol), which were then stirred for a further 24 hours. The reaction was monitored using thin layer chromatography (TLC) using an eluent consisting of a mixture of hexane and ethyl acetate (55:45). After the reaction was finished, the produced reaction mixture was concentrated and washed with 5% hydrochloric acid. The crude product was refined by column chromatography using a mobile phase of hexane:ethyl acetate (55:45) and a stationary phase of silica gel (60-120 mesh). Curcumin-NSAIDs Conjugates were made by collecting, condensing, and drying the various monoconjugated fractions.
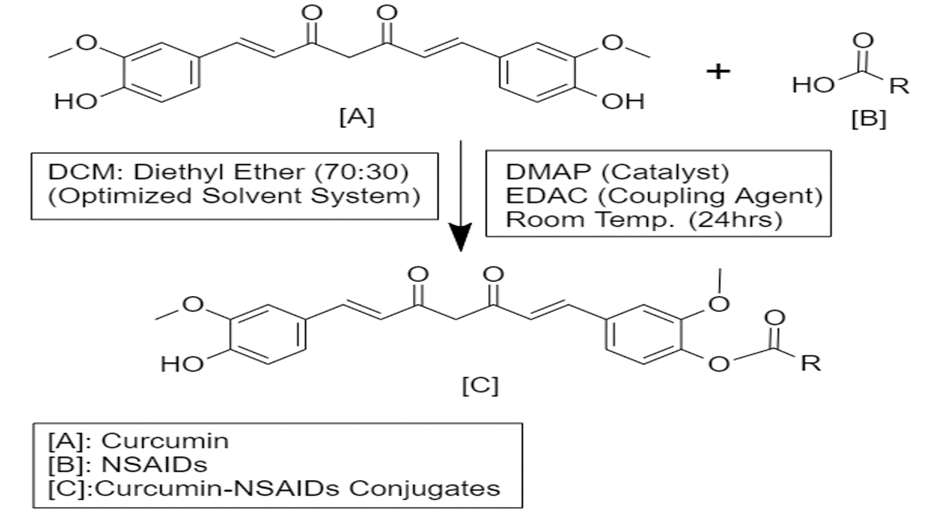
Figure 2: Synthesis of Curcumin-NSAIDs Conjugates
BIOAVAILABILITY
In Sprague-Dawley rats, the bioavailability of curcumin and its NSAID conjugates was measured. By random distribution, the animals were split up into five groups of six each. Curcumin (650 mg/kg) and several solutions containing curcumin-NSAID conjugates (150 mg/kg) in 0.5 percent CMC were administered orally. Blood samples (~0.5 ml) were extracted from the retro-orbital plexus under light ether anesthesia, and they were periodically added to micro centrifuge tubes that had been heparinized (containing 50 l of 1000 U of heparin). The blood samples were centrifuged at 10,000 rpm for 7 minutes at 15° to separate the plasma. Curcumin was extracted using plasma, as was previously indicated. In summary, 50 µl of internal standard (600 ng/ml rhodamine G) was combined with 180 µl of plasma and vortexed for 60 seconds. After adding 220 µl of ACN, the sample was vortexed for an additional two minutes and centrifuged at 10,000 rpm for five minutes. The amount of curcumin in the supernatant was determined using HPLC. As was already described in [29], curcumin was extracted using plasma.
ANTI-INFLAMMATORY ACTIVITY
Male and female adult rats in good health, weighing between 120 and 160 grams, were used for the study. They received water to drink and a daily pellet meal. Six groups of six rats each were created out of the rats. A placebo was administered to one group of animals, and the standard treatment (Naproxen) was given to the other. The test substances were distributed to the remaining groups. Groups (standard, control, and test compound) received oral administration of naproxen and sample compound, respectively, prepared as a 1% CMC suspension. Curcumin was isolated from plasma. The amount was estimated after 30 minutes of subcutaneous injection of 0.1 mL of newly made carrageenan suspension in 0.9 percent NaCl solution in the paw. The foot volume was measured once more at one and two hours, and the mean increase in paw volume in each group was computed. The paw volume was measured with a water plethysmometer. The quantity of oedema that had been created was calculated using the volume differential. The percentage of inhibition value is computed using the formula below:
[1- Dt/Dc]*100
Dt =Paw volumes of oedema in test
Dc==Paw oedema volumes in control.
ANTI-BACTERIAL ACTIVITY
The agar-diffusion technique was used to determine preliminary antibacterial activity. A nutritive agar well diffusion test was carried out with 15 lbs (121°C) pressure and immediately after removal from the autoclave, it was chilled to 50-55°C in the water, as described by Magaliet al. (2004). The cooled medium was poured onto sterile petri plates with a constant 4mm depth, equal to about 40mL and kept for solidification on a 90mm plate. After the medium had solidified, the culture was injected in a laminar air flow on the media. Within 15 minutes of changing the density of the inoculum, a sterile cotton swab was dipped into the normal bacterial suspension or injected with 1mL of organism solution. The sterile swab was used to ensure that the inoculums were evenly distributed throughout the surface of the nutrient agar medium. To ensure adequate moisture absorption, the plates were left intact for 3 to 5 minutes. The 7mm cork borer was sterilised in the preparation of the agar wells, and the diluted test compound solution concentrations of 25, 50, 75, 100 and 200 g/ml in each well were put, with ciprofloxacin controlled at 100%. For bacterial growth, 37°C and 20–25°C incubation plates were incubated in an incubator. Inhibition zones have been discovered in the vicinity of the agar wells. The diameters of the zones were measured in millimetres.
The inhibition ?n be determined using the formula:
Percentage inhibition = (I) diameter of inhibition zone in mm/ (90) diameter of petri plate in mm
ANTI-ULCER ACTIVITY
Wistar albino male rats weighing 200-250 g were split into Six groups (n=6) and given free access to water for two days. The animals were dehydrated for another 24 hours on the third day before undergoing surgery under general anaesthesia with Ketamine/diazepam. Group one animals were used as a typical ulcerative control group, with their pylorus ligated and just saline. The same model was used to ligate the pylorus of the rats in the remaining five groups. The animals in groups were administered 20 mg/kg (dissolved in saline) curcumin, CDC, CAC, CNC and CIC via oral gavage and were decapitated after 19 hours. The excised stomach was then filled with 15 mL of 4 percent formalin after the gastric juice was collected. The fixed stomach was opened along its larger curvature and the ulcer spots were measured, ulcer lesion index and percentage inhibitions were calculated. [30] [31] [32]
RHEUMATOID ARTHRITIS
Balb/c mice with body weights between 15 and 20 g were chosen to create an animal model of antigen-induced arthritis. By injecting modified Streptococcus mutans cell wall extract (SCW) that had already been prepared with proteolytic enzymes intravenously, arthritis was generated. The animals were divided into seven groups, each with six animals, to test the antirheumatic activity of curcumin and curcumin NSAID conjugates. Group 1: normal control; Group 2: disease control; Group 3 oral curcumin 62.50 mg/kg/day; Group 4: oral CAC 107.1 mg/kg/day; Group 5: oral CNC 107.1 mg/kg/day; Group 6: oral CDC 107.1 mg/kg/day; Group 7: oral CIC 107.1 mg/kg/day. Seven days before the induction of arthritis, medication administration began and it continued throughout the duration of the trial. The study lasted for fourteen days. On the eighth day of the trial, 10 mg/kg of a modified SCW extract was intravenously injected to cause arthritis. The test samples were still being given to the animals up to the fourteenth day, during which time they were being watched for changes in joint thickness, tail thickness, body weight and mobility. [33][34][35][36]
RESULT AND DISCUSSION
BIOAVAILABILITY
The plasma concentration vs. time profiles in rats, of curcumin (650mg/kg) following single oral administration of CDC, CIC, CNC, CAC (150 mg/kg), are shown in fig. 3. The pharmacokinetic parameters were estimated by one compartmental analysis of the experimental data. The peak plasma concentration (Cmax) of curcumin obtained was 80±2.41 ng/ml. The mean values of AUC0-? of curcumin were 3.765±0.205 mg/ml/h. CDC exhibited Cmax of 58.93±1.49 ng/ml, mean values of AUC0-? of CDC were 3.840±0.262 mg/ml/h, CIC exhibited Cmax of 57.93±1.79 ng/ml, mean values of AUC0-? of CIC were 4.264±0.224 mg/ml/h, CNC exhibited Cmax of 49.93±1.99 ng/ml, mean values of AUC0-? of CNC were 4.031±0.203 mg/ml/h, CAC exhibited Cmax of 47.93±2.09 ng/ml, mean values of AUC0-? of CAC were 4.044±0.182 mg/ml/h. On comparing the AUC0-? values of curcumin and with other conjugate and it is found that enhancement in bioavailability of curcumin was observed in other conjugate.
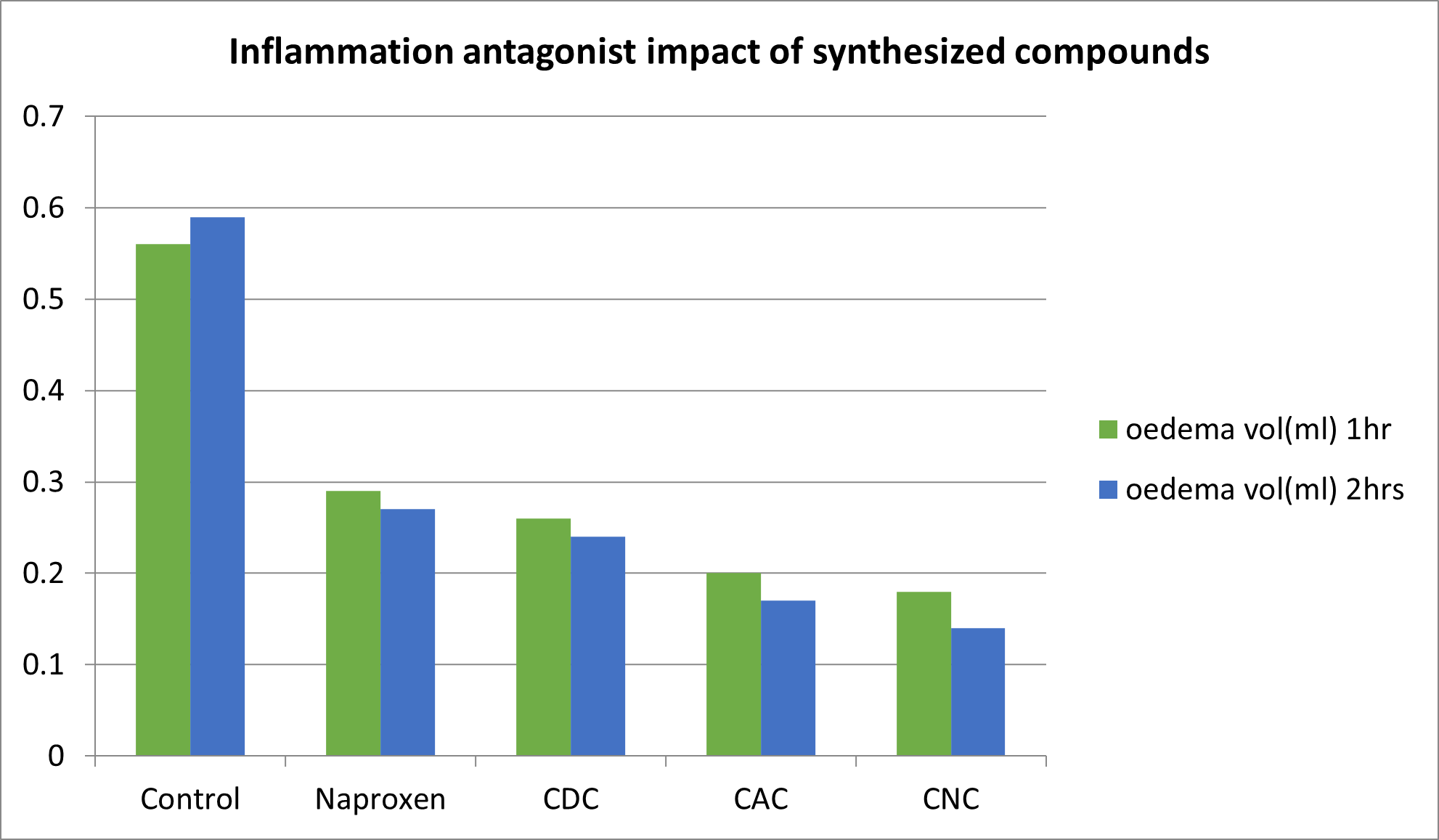
Figure3: Stability studies of curcumin-diclofenac conjugate in different media.
NTI-INFLAMMATORY ACTIVITY
By assessing the % inhibition of oedema with a 1 and 2 hour interval, anti-inflammatory action was discovered. After one hour of delivery, naproxen (48.3%), CDC (53.6%), CAC (64.28%), CNC (67.85%) and CIC were shown to have the highest levels of inhibition (51.78 percent ). After two hours of treatment, it was discovered that naproxen (54.23 percent), CDC (59.32 percent), CAC (71.18 percent), CNC (76.27 percent) and CIC had the highest levels of inhibition (57.62 percent ). The outcomes demonstrate the respective benefits of curcumin conjugates.
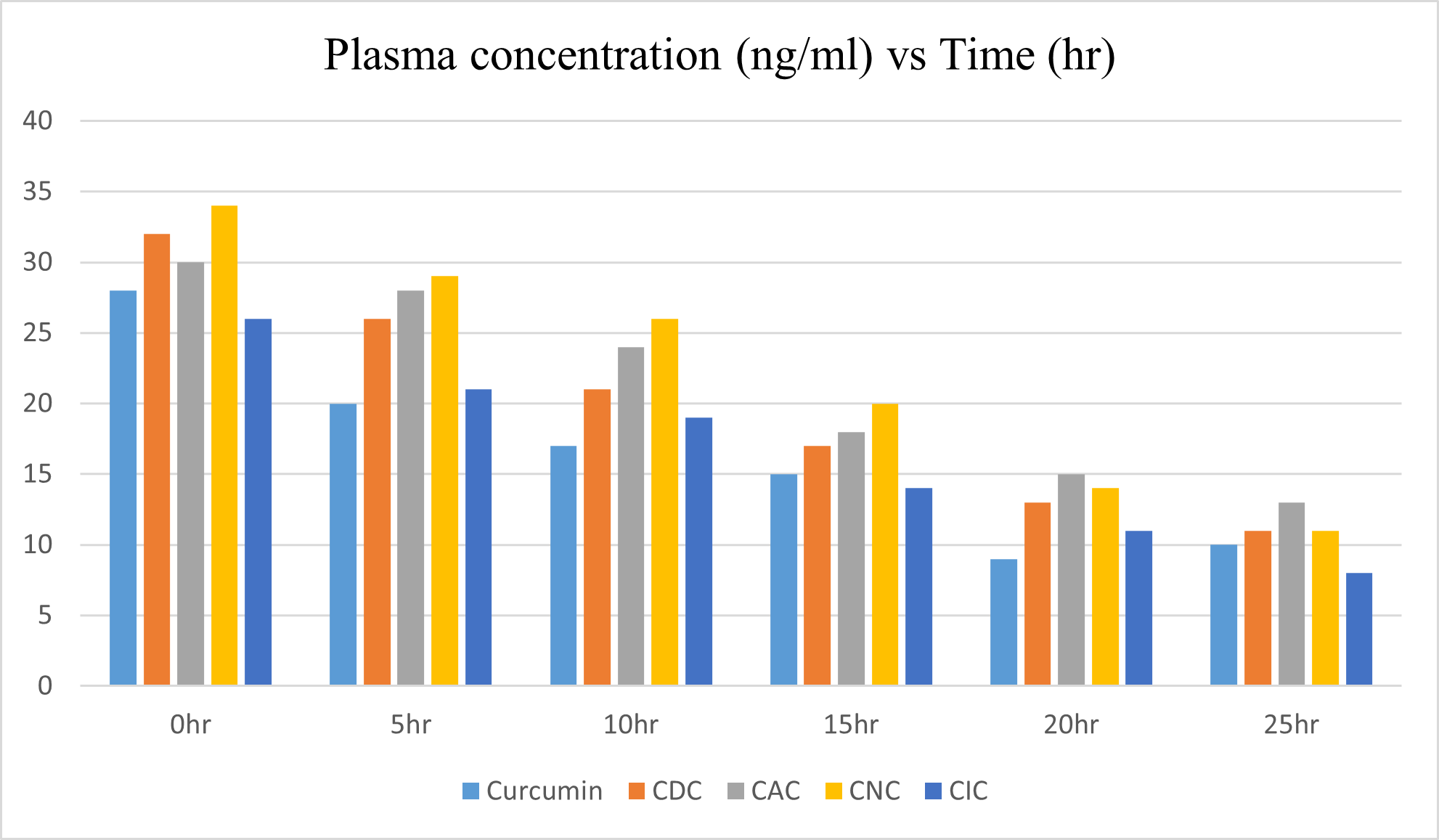
Figure4: Anti-inflammatory effect of synthesized compounds
ANTI-BACTERIAL ACTIVITY
The antibacterial properties of synthesised compounds (CDC, CAC, CNC and CIC) were assessed in vitro against pathogens such gramme positive (Bacillus pumilus, Staphylococcus aureus) and gramme negative (Escherichia coli, Klebsiellapneumoniae). At doses of 1000 g/mL, the bactericidal activity of all four synthesised compounds was assessed. In comparison to the standard (ciprofloxacin), which had a value of 51.94%, the mean percentage of zone inhibition for the CDC was determined to be 54.99%, while it was 58.33% for CAC, 60.83% for CNC and 45.27% for CIC. The aforementioned information demonstrates the curative properties of curcumin conjugates.
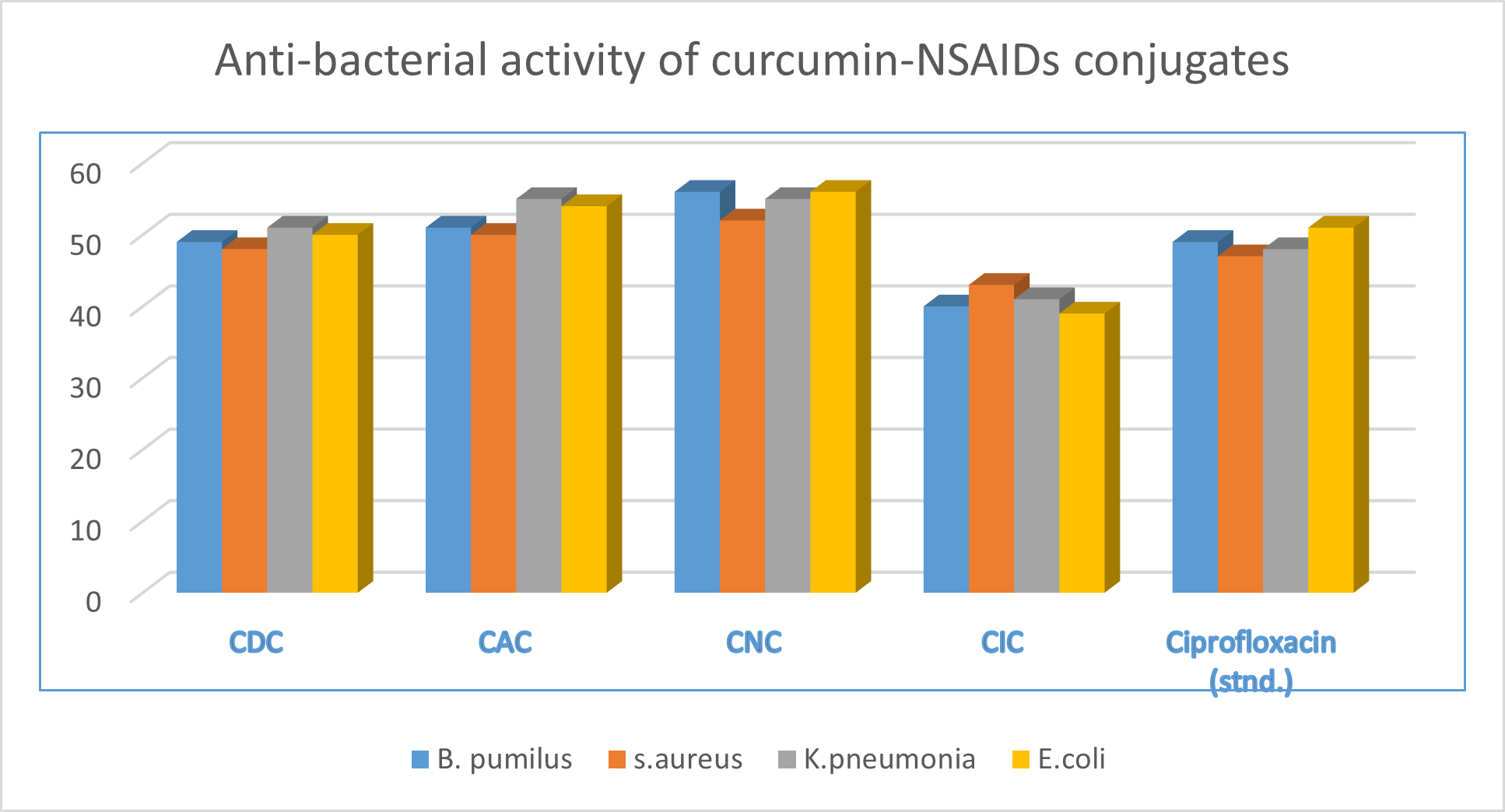
Figure 5 Anti-bacterial effect of synthesized compounds
ANTI-ULCER ACTIVITY
The mean±SE value of gastric ulcer lesion index have been shown as 59.66±3.21 of normal saline, 7.52±1.24 of curcumin, 10.21±1.77 of CDC, 11.28±2.32 of CAC, 14.06±1.88 of CNC and 22.37±2.06 of CIC. The percentage inhibition of ulcerative lesion has been shown in fig7. The above data show significant result. The mean±SE value of the stomach ulcer lesion index was determined to be 59.66±3.21 for normal saline, 7.52±1.24 for curcumin, 10.21±1.77 for the CDC, 11.28±2.32 for the CAC, 14.06±1.88 for the CNC and 22.37±2.06 for the CIC. Figure 7 displays the % inhibition of the ulcerative lesion. The figures up top indicate a substantial outcome.

Figure6 :The percentage inhibition of ulcerative lesion
RHEUMATOID ARTHRITIS
Each animal received a score between 5 and 20 points to represent the degree of arthritis. As a result, the statistics demonstrated how severe arthritis was among various populations. (Table 1).
Table 1: Evaluation of antiarthritic activity of curcumin, diclofenac and synthesized componds in balb/c mice

Conjugation as an important strategy has been adopted resulting in a large number of drug molecules with enhanced solubility, bioavailability and in vivo performance of small and large molecules. In our study, the drug-drug conjugate was adopted as a novel approach to synthesize different Curcumin NSAIDs Conjugates to increase the bioavailability of curcumin, enhance the therapeutic benefit of NSAIDS and in addition, the inherent beneficial effects of curcumin in the GIT has the potential to alleviate the side effects and severity of arthritis treatment. The enhanced bioavailability of curcumin in Curcumin NSAIDs Conjugates was by over many folds can be attributed to the improved stability of Conjugates. Conjugates also exhibited better anti-inflammatory activity in arthritis in Balb/c mice. Thus, conjugation of Curcumin with NSAIDs molecules could be a novel strategy for improvement of its bioavailability and for potentiating its numerous pharmacological activities.
CONFIRMATION OF STRUCTURAL FEATURES OF SYNTHESIZED COMPOUNDS
CDC-Curcumin-Diclofenac Conjugate

Figure 7: CDC-Curcumin-Diclofenac Conjugate
FT-IR Peaks
There were specific IR peaks due to Curcumin at 3508 cm-1 1628 cm-1 1603 cm-1 1510 cm-1 1427 cm-1 and 1278 cm-1 The spectrum of Curcumin-Diclofenac Conjugate showed an infrared band at around 1602.85 cm-1 (C=C); 3387.00 cm-1 (N-H amine) peaks; stretching vibrations due to phenolic hydroxyl groups at 3200–3500 cm-1; stretching vibration at 1490 cm-1 associated with the aromatic C=C bond and a bending vibration at 1246 cm-1 attributed to the phenolic C-O group; a stretching band at 1735 cm-1 due to the C=O ester group.

Figure 8: FT-IR peaks of CDC-Curcumin-Diclofenac Conjugate
1H NMR of compounds
1H NMR (CDCl3): ? 3.75 (s, 3H), 3.94 (s, 3H), 4.09 (s, 2H), 5.87 (d, 2H), 6.47 (d, 2H, J=16.4 Hz), 6.58 (d, 1H, J=8.04 Hz), 6.73 (s, 1H), 6.98 (m, 3H), 7.06 (m, 3H), 7.15 (m, 3H), 7.34 (m, 3H), 7.57 (d, 1H, J=7.2 Hz), 7.61 9d, 1H, J=6.92 Hz)..

Figure 9: 1H NMR peaks of CDC-Curcumin-Diclofenac Conjugate
CAC- Curcumin-Aceclofenac Conjugate

Figure 10: CAC- Curcumin-Aceclofenac Conjugate
FT-IR Peaks
There were specific IR peaks due to Curcumin at 3508 cm-1 1628 cm-1 1603 cm-1 1510 cm-1 1427 cm-1 and 1278 cm-1 . The spectrum of Curcumin-Aceclofenac Conjugate showed infrared band at around 3320 cm -1 (N-H stretching); 3287 cm -1 (O-H stretching); 1715 cm -1 (C=O stretching); 1284 cm -1 (C-N aromatic amine); 1344.38 cm -1 (O-H in plane bending; 3000 cm -1 (C-H Stretching); 1455 cm -1 (Aromatic ring stretch); 1659 cm -1 (C=C stretch); stretching vibrations due to phenolic hydroxyl groups at 3200–3500 cm-1

Figure 11: FT-IR peaks of CAC-Curcumin-Diclofenac Conjugate
1H NMR of compound
1H NMR (CDCl3): ? 3.66 (s, 3H), 3.90 (s, 3H), 4.02 (s, 2H), 5.82 (d, 2H), 6.471(d, 2H, J=16.4 Hz), 6.53 (d, 1H, J=8.04 Hz), 6.68 (s, 1H), 6.88 (m, 3H), 7.08 (m, 3H), 7.17 (m, 3H), 7.36 (m, 3H), 7.59 (d, 1H, J=7.2 Hz)

Figure 12: 1H NMR peaks of CAC-Curcumin-Diclofenac Conjugate
CNC- Curcumin Naproxen Conjugate
Figure 13: CNC-Curcumin-Diclofenac Conjugate
FT-IR Peaks
There were specific IR peaks due to Curcumin at 3508 cm-1 1628 cm-1 1603 cm-1 1510 cm-1 1427 cm-1 and 1278 cm-1 . The spectrum of Curcumin-Naproxen Conjugate showed an intense, well defined peak, infrared band at around at 1252 cm?1 due to C–O stretching; 1583 cm?1 due to COO– stretching; C–C aromatic stretching at 1631 cm?1; C–H aliphatic stretch at 2840 cm?1; stretching vibrations due to phenolic hydroxyl groups at 3200–3500 cm-1; stretching vibration at 1490 cm-1 associated with the aromatic C=C bond and a bending vibration at 1246 cm-1 attributed to the phenolic C-O group; a stretching band at 1735 cm-1 due to the C=O ester group .
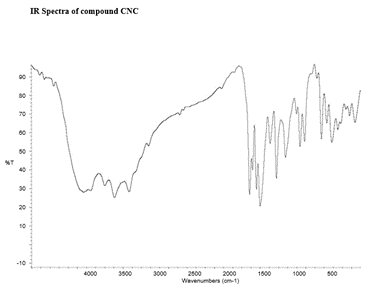
Figure 14: FT-IR peaks of CNC-Curcumin-Diclofenac Conjugate
1H NMR of compounds
1H NMR (CDCl3): ? 3.70 (s, 3H), 3.92 (s, 3H), 4.11 (s, 2H), 5.83 (d, 2H), 6.40 (d, 2H, J=16.4 Hz), 6.51 (d, 1H, J=8.04 Hz), 6.70 (s, 1H), 6.94 (m, 3H), 7.04 (m, 3H), 7.10 (m, 3H), 7.31 (m, 3H), 7.56 (d, 1H, J=7.2 Hz)

Figure 15: 1H NMR peaks of CNC-Curcumin-Diclofenac Conjugate
CIC- Curcumin-Ibuprofen Conjugate

Figure 16: CIC-Curcumin-Diclofenac Conjugate
FT-IR Peaks
There were specific IR peaks due to Curcumin at 3515 cm-1 1632 cm-1 1598 cm-1 1510 cm-1 1427 cm-1 and 1278 cm-1 The spectrum of Curcumin-Naproxen Conjugate showed an infrared band at 1252 cm?1 due to C–O stretching; 1580 cm?1 due to COO– stretching; C–C aromatic stretching at 1634 cm?1; C–H aliphatic stretch at 2840 cm?1; stretching vibrations due to phenolic hydroxyl groups at 3210–3500 cm-1; and a bending vibration at 1246 cm-1 attributed to the phenolic C-O group; a stretching band at 1735 cm-1 due to the C=O ester group.

Figure 17: FT-IR peaks of CIC-Curcumin-Diclofenac Conjugate
1H NMR of compounds
1H NMR (CDCl3): ? 3.69 (s, 3H), 3.88 (s, 3H), 3.99 (s, 2H), 5.80 (d, 2H), 6.02 (d, 2H, J=16.4 Hz), 6.58 (d, 1H, J=8.04 Hz), 6.71 (s, 1H), 6.91 (m, 3H), 7.06 (m, 3H), 7.08 (m, 3H), 7.31 (m, 3H), 7.50 (d, 1H, J=7.2 Hz)

Figure 18: 1H NMR peaks of CIC-Curcumin-Diclofenac Conjugate
CONCLUSION:
Many pharmacological compounds with improved solubility, stability, permeability, bioavailability, organoleptic characteristics and in vivo performance have been produced as a result of the essential method known as conjugation. Drug-drug conjugates were used in our study as a novel approach to synthesise CDC, CIC, CNC and CAC in order to increase the bioavailability of curcumin, improve the therapeutic benefit of diclofenac, and in addition, the inherent beneficial effects of curcumin in the GIT have the potential to reduce the side effects and severity of arthritis treatment. The improved stability of CDC, CIC, CNC and CAC as well as their affinity for the enzymes attached to the gut can be related to the increased bioavailability of curcumin in other conjugates. Furthermore, they demonstrated superior anti-inflammatory efficacy in mice used in a modified SCW model of arthritis. Additionally, they demonstrated strong antibacterial and antiulcer action. Therefore, conjugating curcumin with medicinal molecules may be a creative way to increase its bioavailability and enhance its wide range of pharmacological effects
REFERENCES
- Goel A, Kunnumakkara AB, Aggarwal BB. Curcumin as “Curcumin”: From kitchen to clinic. BiochemPharmacol. 2008;75:787–809.
- Duvoix A, Blasius R, Delhalle S, Schnekenburger M, Morceau F, Henry E, et al. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005;223:181–90.
- Jayaprakasha G, Jaganmohan Rao L, Sakariah K. Antioxidant activities of curcumin, demethoxycurcumin and bisdemethoxycurcumin. Food Chem. 2006;98:720–4.
- Kohli K, Ali J, Ansari M, Raheman Z. Curcumin: A natural antiinflammatory agent. Indian J Pharmacol. 2005;37:141.
- Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: Problems and promises. MolPharmaceut. 2007;4:807–18.
- Ireson CR, Jones DJ, Orr S, Coughtrie MW, Boocock DJ, Williams ML, et al. Metabolism of the cancer chemopreventive agent curcumin in human and rat intestine. Cancer EpidermBiomar. 2002;11:105–11.
- Vareed SK, Kakarala M, Ruffin MT, Crowell JA, Normolle DP, Djuric Z, et al. Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects. Cancer Epidemiol Biomarkers Prev. 2008;17:1411–7.
- Agrawal DK and Mishra PK. Curcumin and its analogues: potential anticancer agents. Med Res Rev.30; 2010; 818-860.
- Itokawa H, Shi Q, Akiyama T, Morris-Natschke SL and Lee KH. Recent advances in the investigation of curcuminoids. Chin Med. 17; 2008; 3-11.
- Dastur JF. Medicinal Plants of India and Pakistan. Treasure House of Books. Bombay. 1970.
- Haddad M, Sauvain M and Deharo E. Curcuma as a parasiticidal agent: a review. Planta Med. 77;2011; 672-678.
- Zhu ZY. Flora Sichuanica. Sichuan Nationalities Publishing House. 1992; pp. 604-610.
- Ravindran PN, Ravindran K, Nirmal B and Sivaraman K. Turmeric: the genus Curcuma; Medicinal and aromatic plantsindustrial profiles. CRC Press, Taylor and Francis Group Boca Raton, FL. 2007; pp. 236.
- Khanna NM. Turmeric-nature’s precious gift. Current Sci. 76(10);1999:1351-1356.
- Grykiewicz, G.; Silfirski, P. Curucmin and curcuminoids in quest for medicinal status. ActaBiochim. Pol. 2012, 59, 201–212
- Esatbeyoglu, T.; Huebbe, P.; Insa, M.A.; DawnChin, E.; Wagner, A.E.; Rimbach, G. Curcumin—From Molecule to Biological Function. Angew. Chem. Int. Ed. 2012, 51, 5308–5332.
- Gupta, S.; Prasad, S.; Ji, H.K.; Patchva, S.; Webb, L.J.; Priyadarsini, K.I.; Aggarwal, B.B. Multitargeting by curcumin as revealed by molecular interaction studies. Nat. Prod. Rep. 2011, 28, 1937–1955.
- Priyadarsini, K.I. Chemical and structural features influencing the biological activity of curcumin. Curr. Pharm. Des. 2013, 19, 2093–2100
- Priyadarsini, K.I. Photophysics, Photochemistry and Photobiology of Curcumin: Studies from organic solutions, bio-mimetics and living cells. J. Photochem. Photobiol. C 2009, 10, 81–96.
- AGGARWAL, B.B., A. KUMAR & A.C. BHARTI. 2003. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 23: 363–398.
- Durga M. Arvapalli, Alex T. Sheardy, KokouganAllado, Harish Chevva, Ziyu Yin, Jianjun Wei. Design of Curcumin Loaded Carbon Nanodots Delivery System: Enhanced Bioavailability, Release Kinetics and Anticancer Activity. ACS Applied Bio Materials 2020, 3 (12) , 8776-8785.
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises: Full Text Finder Results. Mol. Pharm. 2007, 4, 807–818.
- Heger, M.; van Golen, R.F.; Broekgaarden, M.; Michel, M.C. The Molecular Basis for the Pharmacokinetics and Pharmacodynamics of Curcumin and Its Metabolites in Relation to Cancer. Pharmacol. Rev. 2013.
- Serafini, M.M.; Catanzaro, M.; Rosini, M.; Racchi, M.; Lanni, C. Curcumin in Alzheimer’s disease: Can we think to new strategies and perspectives for this molecule? Pharmacol. Res. 2017, 124, 146–155.
- Adiwidjaja, J.; McLachlan, A.J.; Boddy, A.V. Curcumin as a clinically-promising anti-cancer agent: Pharmacokinetics and drug interactions. Expert Opin. Drug Metab. Toxicol. 2017, 13, 953–972.
- van den Bekerom MPJ, Sjer A, Somford MP, Bulstra GH, Struijs PAA, Kerkhoffs GMMJ. Non-steroidal anti-inflammatory drugs (NSAIDs) for treating acute ankle sprains in adults: benefits outweigh adverse events. Knee Surg Sports TraumatolArthrosc. 2015 Aug;23(8):2390-2399.
- Scheiman, James M.; Yeomans, Neville D.; Talley, Nicholas J.; Vakil, Nimish; Chan, Francis K. L.; Tulassay, Zsolt; Rainoldi, Jorge L.; Szczepanski, Leszek; Ung, Kjell-Arne; Kleczkowski, Dariusz; Ahlbom, Henrik; Næsdal, Jørgen; and Hawkey, Christopher; Prevention of Ulcers by Esomeprazole in At-Risk Patients Using Non-Selective NSAIDs and COX-2 Inhibitors; American Journal of Gastroenterology: April 2006 - Volume 101 - Issue 4 - p 701-710.
- Sabzwari SR, Qidwai W, Bhanji ;Polypharmacy in elderly: a cautious trail to tread., J Pak Med Assoc. 2013 May; 63(5):624-7.
- Sintara K, Thong-Ngam D, Patumraj S, Klaikeaw N, Chatsuwan T. Curcumin suppresses gastric NF-?B activation and macromolecular leakage in Helicobacter pylori-infected rats. World J Gastroenterol. 2010;16:4039
- Saroj K. Pal, Pulok K. Mukherjee and B. P. Saha, Studies on the Antiulcer Activity of Moringa oleifera Leaf Extract on Gastric Ulcer Models in Rats, PHYTOTHERAPY RESEARCH, VOL. 9,463-465 (1995)
- Pillai, N. R. and Santhakumari, G. (1984). Effects of nimbidin on acute and chronic gastroduodenal ulcer models in experimental animals. flanta Med. 50, 143-146.
- Aguwa Cletus, N. and Ramanujam, T. R. (1984). Antiulcer effects of trimipramine using various laboratory models. Jpn J. Pharmacol. 36, 125-129
- S. K. Jain, M. S. Gill,1 H. S. Pawar and Sarasija Suresh* Novel Curcumin Diclofenac Conjugate Enhanced Curcumin Bioavailability and Efficacy in Streptococcal Cell Wall-induced Arthritis, Indian J Pharm Sci. 2014 Sep-Oct; 76(5): 415–422.
- Li X, Bradford BU, Dalldorf F, Goyert SM, Stimpson SA, Thurman RG, et al. CD14 mediates the innate immune responses to arthritopathogenic peptidoglycan-polysaccharide complexes of Gram-positive bacterial cell walls. Arthritis Res Ther. 2004;6:R273–81.
- Kannan K, Ortmann RA, Kimpel D. Animal models of rheumatoid arthritis and their relevance to human disease. Pathophysiol. 2005;12:167–81.
Koga T, Kakimoto K, Hirofuji T, Kotani S, Ohkuni H, Watanabe K, et al. Acute joint inflammation in mice after systemic injection of the cell wall, its peptidoglycan, and chemically defined peptidoglycan subunits from various bacteria. Infect Immun. 1985;50:27–34.


 Lovy Sharma
Lovy Sharma



















 10.5281/zenodo.13337214
10.5281/zenodo.13337214