|
Chromatographic Conditions
|
Wavelength
|
Sample
|
Reference
|
|
Methanol: Acetonitrile: Phosphate Buffer (45:45:10)
|
246nm
|
-
|
(7)
|
|
Column C18, 0.005 mm,
mobile phase - ammonia acetate, ACN, and Tetrahydrofuran buffer (pH 4) (25:70:5 %v/v),
Pump rate- 1000 µL/min
|
The detection wavelength was set at 248 nanometres
|
Tablet
|
(8)
|
|
Stationary phase- 15 cm × 4.6 cm, 0.005 mm,
Eluent – A solution prepared using 30% methanol and 70% of the second solvent (by volume), with the solution maintained at pH 4.
|
247 nm
|
Plasma
|
(9)
|
|
Column C18, 15 cm × 0.46 cm, 0.005 mm,
Mobile phase - 40:60 acetonitrile: aqueous solution of hydrogen tetrabutylammonium sulfate regulated to pH value of 3,
Pump rate- 1000 µL/min
|
386 nm
|
Plasma
|
(10)
|
|
C18 used as stationary phase with dimensions 15 cm × 0.46 cm, 0.005 mm,
Mobile phase - ACN: 30 mM phosphate buffer (pH 2.9) (55:45 %v/v),
Pump flow- 1000 µL/min
|
240 nm
|
Tablets
|
(11)
|
|
Stationary phase- Column C18,
Mobile phase- Water: ACN (48:52 %v/v) pH 2 Adjusted with 80% O-phosphoric acid)
|
245 nm
|
Tablets
|
(12)
|
|
C8, Mobile phase - phosphate buffer: ACN (pH 2.3), Pump rate- 1000 µL/min,
Volume of injection: 5 μL, Temperature: 60°C
|
210 nm
|
Tablets
|
(13)
|
|
C18 column 25 cm × 0.46 cm, 0.005 mm,
Mobile phase: 50% of 10 mM aqueous solution of ammonium salt of acetic acid, adjusted to pH value of 3: 50% of ACN(by volume)
Pump rate- 1000 µL/min
|
254 nm
|
Pharmaceutical formulations
|
(14)
|
|
Stationary phase- C18, 15 cm × 0.46 cm, 0.005 mm, Temperature: 20-25 °C,
Eluent- 70% of ACN: 30% of Aqueous acetic acid at 0.001 fraction by volume.
Pump flow- 1000 µL/min, Volume of injection -10 μL.
|
246 nm
|
Plasma
|
(15)
|
|
Column - C18, 0.46 cm × 7.5 cm, 0.0035 mm,
Eluent- 70% of ACN: 30% of Aqueous formic acid at 0.001 fraction by volume.
Pump flow- 1000 µL/min,
Volume of injection- 0.01 mL
|
Absorbance recorded at 238 nanometres
|
Tablet solid dosage form
|
(16)
|
|
Octadecyl (ODS) column 25 cm × 0.46 cm, 0.005 mm,
Mobile phase- sodium phosphate solution at a 0.05 M concentration with methanol in a 3:7 volume ratio and finely tuning the mixture’s pH to 4.1 using orthophosphoric acid as the acidifying agent.,
Thermal conditions: 25 ± 0.5°C, Pump rate- 1000 µL/minutes
|
247 nm
|
Bulk,
tablets
and nanoemulsion
|
(17)
|
|
Column - C18 25 cm × 0.46 cm,
Eluent- 70% Methanol and 30% acetate aq. Solution by volume, with the pH adjusted to 4.1 through the addition of orthophosphoric acid,
Pump flow- 1000 µL/min and Volume of injection- 20 μL
|
245 nm
|
Tablets
|
(18)
|
|
Column - C18, 25 cm × 0.46 cm, 0.005 mm,
Pump rate- 1000 µL/min,
Mobile Phase- 53.55% of ACN : 05% of Methyl alcohol: 41.45% of 0.001 fraction of TEA adjusted to pH 3 using O- phosphoric acid as the acidifying agent
|
220 nm
|
Human
plasma
|
(19)
|
|
Column- C18, 15 cm × 0.46 cm, 0.005 mm,
Eluent- 68% methyl alcohol: 32% of water by volume; pH meticulously adjusted to 3 through addition of TFA,
Pump rate- 1500 µL/min
|
241 nm
|
Human
serum
|
(20)
|
|
Column - C18, 15 cm × 0.46 cm, 0.005 mm,
Eluent- 43% of 10% methyl alcohol in 0.05 M sodium phosphate adjust solution pH to 3.5: 57% of methyl alcohol by volume,
Pump rate- 1200 µL/min
|
247 nm
|
Plasma
|
(21)
|
|
Column - C18, 0.46 cm × 15 cm, 0.005 mm, Mobile phase- 45% of ACN and 55% of a solution containing 25 mM of potassium dihydrogen orthophosphate, maintained at an acidity level of pH 5, Pump rate- 1500 µL/min
|
246 nm
|
Nanocrystals
|
(22)
|
|
Column - C18, 25 cm × 0.46 cm, 0.005 mm,
Eluent- 60% of ACN: 40% of phosphate buffer solution by volume, maintained at pH 3, Pump flow- 1000 µL/min,
Volume of injection- 25 μL
|
235 nm
|
Bulk and tablets
|
(23)
|
|
Column - C18, 0.005 mm, Mobile phase- 0.1 % acetic acid: ACN (45:55 v/v) pH 3.8
Pump rate – 800 µL/min
|
246 nm
|
Tablets
|
(24)
|
|
Column - ODS-AQ YMC 5 cm × 0.46 cm, 0.003 mm, Eluent- Ethanol: formic acid (pH 2.5) (50:50 v/v) Pump rate- 1000 µL/min,
Column oven temperature- 40°C
|
238 nm
|
Tablets
|
(25)
|
|
Stationary phase- C18, 250 mm × 4.6 cm, 0.005 mm,
Eluent– 35% of solution containing phosphate maintained at pH 4.5: 65% of CAN by volume, Pump flow- 1000 µL/min
|
228 nm
|
Pharmaceutical dosage form
|
(26)
|
|
Stationary phase- Octadecyl functionalized separation medium, 25 cm x 0.46 cm, 0.005 mm, Eluent- 60% of 20 mM sodium acetate maintained at pH 4: a solvent system consisting of acetone, triol and methanol each making upto 20% of the total volume.
Pump rate- 700 µL/min, Column oven temperature- 40 ºC
|
210 nm
|
Plasma
|
(27)
|
|
Column C18, 15 cm × 0.46 cm, 0.005 mm, Eluent- ACN: methyl alcohol: 20 mM solution of Dipotassium hydrogen phosphate maintained at pH 3 (34.27: 20: 45.73 %v/v)
Pump rate- 2000 µL/min.
|
239 nm
|
Tablet
|
(28)
|
|
Column - Inertsil ODS-3, 0.46 cm x 25 cm, Eluent- water: acetonitrile: Tetrahydrofuran (11:8:6), Thermal conditions- 30°C,
Pump flow- 1000 µL/min,
Volume of injection- 0.01 mL
|
244 nm
|
Tablets
|
(29)
|
|
Column- C18, 150 mm × 4.6 mm, 0.0035 mm, Acetonitrile–water (85:15) as mobile phase adjusted with phosphoric acid at pH 4.5
Injection amount- 0.02 mL,
Pump flow- 1000 µL/min, Thermal conditions- 18-degree Celsius
|
261 nm
|
Tablets
|
(30)
|
|
Stationary phase- C18 15 cm × 0.46 cm, 0.005 mm, Eluent- 0.001 fraction containing phosphoric acid solution pH maintained at 3 and ACN,
Column oven thermal condition- 30 degree Celsius
|
227 nm
|
Human plasma
|
(31)
|


 Pradnya Kumbhar*
Pradnya Kumbhar*
 Dr. Sandeep Waghulde
Dr. Sandeep Waghulde
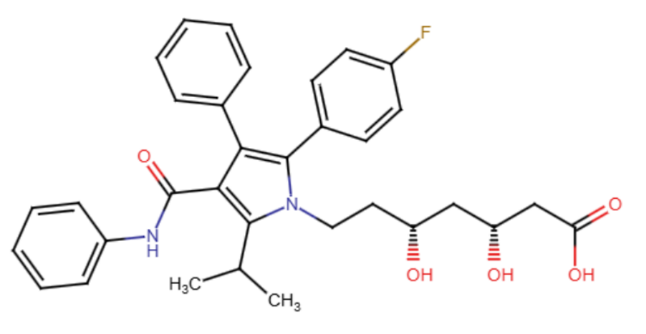
 10.5281/zenodo.18172678
10.5281/zenodo.18172678