A simple, sensitive, rapid and accurate visible spectrophotometric method has been developed for the estimation of famciclovir in pharmaceutical formulations. The proposed method is based on the ion pair complex formation of famciclovir with dyes wool fast blue followed by their extraction with chloroform at 1.5 pH. The absorbance of organic layer was measured for the above complexes at 580 nm. The results of the analysis were validated statistically and were found to be satisfactory.
Famciclovir, Wool Fast Blue, Chloroform, Visible Spectroscopy.
Chemically, famciclovir is known as 2-[2-(2-amino-9H-purin9-yl)ethyl]-1,3-propanediol diacetate. It is a novel antiviral drug, which is highly efficient in the treatment of acute uncomplicated herpes zoster1 and ophthalmic zoster2. Famciclovir is synthetic guanine derivative, which is metabolized to the potent antiviral compound penciclovir. Literature survey reveals that the famciclovir was estimated by using various methods which includes Spectrophotometric method1-12, RP-HPLC method13-15 LC method16-17. The authors developed visible spectrophotometric method for the determination of famciclovir in bulk and in pharmaceutical formulations. In the present work, the reactions of famciclovir treated with wool fast blue and the resultant solution was extracted with chloroform. The extractable ion pair complex was used for estimation of famciclovir.
MATERIALS AND METHODS
Instrumentation
Spectronic 1000 plus UV Visible Spectrophotometer with 1 cm matched quartz cells was used for all spectral and absorbance measurements. AR grade chemicals are used for preparation of reagents and solutions in the present investigations. AR grade methanol and chloroform are used as solvent for determination of famciclovir in pharmaceutical formulations.
Buffer solution (pH 1.5): Buffer solution is prepared by mixing 289 ml of glycine solution (37.52 gm of glycine and 29.24 gm of NaCl are dissolved in 500 ml of distilled water) with 711 ml of 0.1M HCl and pH of the solution is adjusted to 1.5.
Wool Fast Blue solution (0.2% w/v): Wool Fast Blue solution is prepared by dissolving 200 mg of wool fast blue (Fluka) in 100 ml of distilled water.
Standard famciclovir solution: 50 mg of pure famciclovir is dissolved in methanol and the volume is adjusted to 50 ml with methanol. The stock solution is further diluted to get working concentration of 100 mg/mL.
Preparation of Calibration Curve:
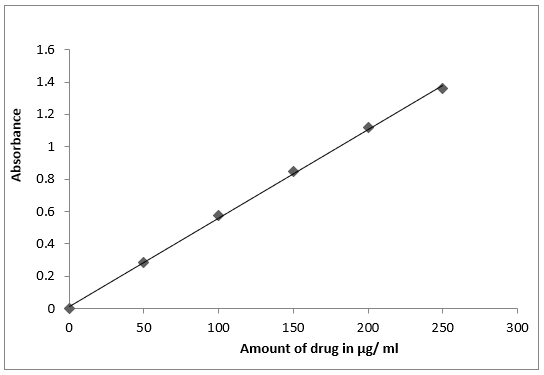
Various aliquots of the standard famciclovir solution ranging from 0.5-2.5 ml are transferred into a series of separating funnel. To each flask, 1.0 ml of wool fast blue solution, 1.0 ml of buffer solution and 5 ml of chloroform are added. Reaction mixture in each funnel is shaken gently for 5 min and allowed to stand for 5 min so as to separate aqueous and chloroform layer. The chloroform layer is separated out and absorbance is measured at 580 nm, against the reagent blank prepared. Calibration graph is obtained by plotting absorbance values against the concentration of famciclovir solution. The calibration curve is found to be linear over a concentration range of 50 to 250 mg/ml of famciclovir. The amount of famciclovir present in the sample is estimated from the calibration graph. The results are presented in fig.1
Pharmaceutical Formulations
For analysis of tablet formulation, twenty tablets of famciclovir are weighed accurately and finely powdered. An accurately weighed portion of powdered sample, equivalent to 50 mg of famciclovir was taken in a 50 ml volumetric flask containing 25 ml of chloroform, sonicated for 20 minutes. The resultant solution is filtered through Whatman filter paper No. 41 into another 50 ml volumetric flask. The filter paper was washed several times with chloroform. The washings were added to the filtrate and the final volume was made up to the mark with methanol. Further sample solution is diluted and treated as per the procedure of the calibration curve. Amount of the drug present in sample was computed from calibration curve. The results are present in table.2.
RESULTS AND DISCUSSION:
The famciclovir treated with Wool fast blue. The resultant solution is extracted with chloroform at 1.5 pH. The ion pair complex was form in extractable chloroform layer. The absorbance of the extractable ion pair complex is measured at 580 nm against the reagent blank. The calibration curve was linear over the range of 50-250 μg/mL of famciclovir. The optical characteristics of the proposed method such as absorption maxima, Beer´s law limits, molar absorptivity and Sandell´s sensitivity are presented in Table 1. The molar absorptivity and Sandell´s sensitivity values shows sensitivity of the method. The regression analysis using method of least squares was made for the slope (b), intercept (a) and correlation (r) obtained from different concentrations and results are summarized in the Table 5.1. The value of correlation coefficient was 0.999, which indicated the good linearity of calibration lines. The percent relative standard deviation calculated from the five measurements of famciclovir shown in Table2. The % RSD is less than 2, which indicates that the method has good reproducibility. The values of standard deviation are low, indicates high accuracy and reproducibility of the method. The‘t’ calculated values are compares well with the theoretical value of 2.78 there by indicating that the precision of the method. There is no effect of additives and excipients such starch, calcium lactose and glucose in the concentrations those present in general pharmaceutical preparations. The proposed method is found to be simple, precise, accurate and time saving, reproducible and can be conveniently adopted for routine analysis of estimation of famciclovir in bulk drugs samples and pharmaceutical formulations as seen from the agreement of the amount of famciclovir in the present method and the labeled amount of the pharmaceutical preparation.
Table 1- Optical Characteristics of The Proposed Method
|
Parameters
|
Proposed Method
|
|
λmax (nm)
|
590
|
|
Beer’s law limit (µg/ml)
|
50-250
|
|
Molar absorptivity (l mole-1 cm-1)
|
1.7x103
|
|
Sandell’s sensitivity
(µg cm-2 / 0.001 absorbance unit)
|
0.1785
|
|
Regression equation (Y = a + bx)
|
Y=0.0055x+0.0153
|
|
Slope (b)
|
0.0055
|
|
Intercept (a)
|
0.0153
|
|
Correlation coefficient (r)
|
0.999
|
*Y = a+bx, where Y is the absorbance and X concentration in μg/ml Table.2-Assay of Famciclovir in Pharmaceutical Formulations


 Dr. Sravanthi Chittela*
Dr. Sravanthi Chittela*
 J. Sudhakar Reddy
J. Sudhakar Reddy
 10.5281/zenodo.15038444
10.5281/zenodo.15038444