Abstract
One of the most sophisticated families of drug deliver methods is liposomal nanomedicine (LNMs), which are therapeutic compounds encapsulated in liposomal nanoparticles (LNs). Several of these LNMs are presently available for purchase, and many more are undergoing clinical testing. Antisense oligonucleotides, small-interfering RNA [siRNA], and plasmid DNA carrying gene therapies are only a few examples of the innovative genetic and conventional medications produced over the last twenty years and their many encapsulation techniques. Among the unique characteristics associated with such LNs are their large (> 6 hours) circulation lifespan, high drug-to-lipid ratio, larg diameter (centered about 100 nm), and remarkable retention of the encapsulated medication. A spherical artificial membrane made of phospholipid bilayers is called a liposome (Fig. 1). Liposomes have drawn a lot of interest as a drug carrier because of their stability, selectivity, biocompatibility, and membrane-like characteristics. Lipomers are spherically-enclosed, lipid-primarily membrane vesicles. Therapeutics abound in lipid nanoparticles, which may or may not have an encapsulated bilayer. Most of those that have received clinical approval have sizes between 50 and 300 nm. Therapeutic proteins have a high selectivity and bio potency and are well-tolerated bioactive molecules employed in various therapeutics. However, they might also exhibit physicochemical instability, impairing therapies by causing a lack of bioactivity. To show how versatile lipid nanoparticles are, this review will give a rundown of their applications in therapeutic protein delivery. The manufacturing method used to create lipid nanoparticles loaded with therapeutic proteins are the subject of particular attention.
Keywords
Film hydration, Microfluidic channel, glycerol phospholipid, Cholesterol, Lipomer Nanoparticles.
Introduction
The use of materials at the nanoscale level for a variety of purposes is known as nanotechnology. A substance at the nanoscale, which is one billionth of a meter, most notably includes DNA with a single strand that has a diameter of around 2.5 nm. Since proteins have a diameter of around 10 nm and DNA possesses a diameter of about 2 nm, tiny particles with size-inspired properties are vital in numerous areas of nutrition for humans. Researchers have been able to examine the optimal personalization of NM traits or biological elements since 2015, thanks to a novel field approach which has sparked discussions among scientists and generated a worldwide market worth more than USD 1.5 trillion. This was set in motion by the original concepts put out by Nobel laureate Richard Feynman in 1960 (Badya, 2020). In addition, Kroto et al.'s 1985 discovery of fullerene C60 expanded the breadth and improved the study of nanotechnologies (Badya, 2020). Additionally, Kroto et al.'s 1985 discovery of fullerene C60 expanded the breadth and improved the area of nanotechnology (Badya, 2020). Inorganic, organic, and dendrimer nanoparticles may be synthesized in various shapes and sizes, including 1D, 2D, and 3D architectures. The electronically limited dimensionality of those particles allows them to be bent, forming sheets, wires, rods, and even particles themselves. Many approaches have been used to produce, and process engineered nanostructures with specific band structures and surface locations for various medicinal purposes. Chemical nanoparticles (CNTs), semiconductors, and two-dimensional carbon materials (TiO2, ZnO, CuO, etc.) enhance the effectiveness and safety of medicinal therapies. Potential medicinal uses, as shown schematically in Figure 1.
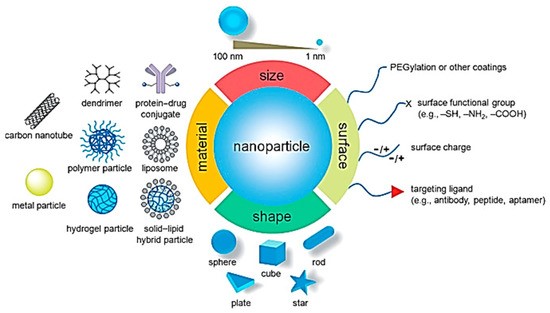
Figure 1: Potential Medicinal Uses of Nanoparticles.
Table 1. Applications of various nanomaterials in the field of medicine
|
Types of Nanomaterial’s
|
Applications
|
|
Metal/metal-oxide nanoparticles
|
Enhanced drug loading and releasing action, permeation characters, Carriers or agents for MRI and ultrasound image Used in apoptosis and angiogenesis.
|
|
CNT
|
Diagnosis in DNA transformation biomarker for changes in protein structure
|
|
Nanocrystals
|
Enhanced soluble drug formulation
|
|
Nanocore shells
|
Contrast imaging for tumors
|
|
1D, 2D nanostructures
|
Accurate throughput scanning Detector for protein diseases Detection of DNA mutation Diagnosis of gene mutation
|
|
0D (quantum dots)
|
Diagnosis of gene and protein structures due to
optical properties
Detection of tumor and lymph nodes
|
? This new field has the potential to create ecologically friendly systems that are vital for reducing negative effects, with natural systems serving as models for how to function with renewable resources. Their optical, electrical, and architectural components remain unreplicated, among additional significant concerns. We have started to consider fresh theoretical models to represent nature and aspects at them nanoscale. The development of the light microscope, which completely altered perception, is the best example. Even if it was created some 500 years ago for observational reasons,changes in the field of technology are still evident today. This perspective has impacted scientists, engineers, and architects on bacteria, diatoms, and cells. As demonstrated in Figure 2, selected biologically generated substances have improved the bottom-up approach to supermolecular structure. These nanometer-to-micrometer substances possess evolving characteristics, usefulness, as well as complexity.
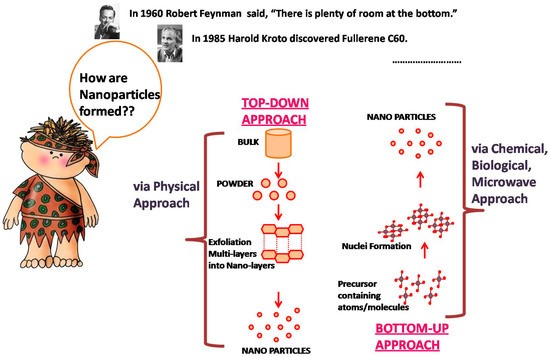
Figure 2: Bottom–Up Approach to Supermolecular Structure.
The molecular machinery method to synthetic self-assembly has also been demonstrated by nature. Bioactive, self-assembling peptide nanofiber hydrogels for tissue regeneration have been the topic of current study in a variety of fields, as reported by reports (Lu, 2018). Sleyter as well as colleagues used periodic crystalline proteins (from B. sphaericus) as a building block for S-layer proteins construction, similar with the based on bottom- technique (Hynonen, 2013) (Pump, 2008) (Sleytre, 2014) (Messner, 1986). Chemical, electrical, optical, and magnetic devices of the next generation may be built using these promising components. However, Allar's group recently reported self-assembly as a method for producing structured materials through covalent and non-covalent interactions (Allara, 2005) (Allara, 1985)
Advantages (A., 2012) (S., 1997) :
• Long-term release.
• Non-ionic
• Site-specific or targeted medication delivery is accomplished by liposomes.
• Preserving drug entrapped in harsh surroundings.
• Able to transport drugs soluble in lipids as well as water.
• It is possible to prevent oxidation of the biodegradable medication.
• Boost the stability of proteins.
• Hydration under control.
• Various pathways for administration are possible.
•Modify both the pharmacodynamics and pharmacokinetics.
• Liposomes offered a number of benefits for introducing genes into cells.
• The chemicals that make up liposomes are positively and negatively charged.
• Liposomes provide DNA with defense against deteriorating agents.
• Huge DNA strands may be carried by liposomes, perhaps in the form of a chromosome.
• Particular cells or tissues are the target of liposomes.
• Liposomes are a more potent and useful drug indicator.
• Liposomes assist in lowering the hazardous drug's exposure to delicate tissue.
• The ability to link quickly with ligands specific to a certain location to produce active targeting.
Disadvantages [9, 10]:
• There is a high production cost.
• Drug and/or molecular encapsulation leakage and/or fusion.
• Phospholipids can occasionally experience processes akin to hydrolysis and oxidation.
• Minimal solubility.
• Minimum half-life.
• A reduced number of stables.
• Rapid absorption by cells of R.E.S.
• Variations between batches.
• It is challenging to manufacture and sterilize liposomes on a big scale; once administered, they cannot be taken out.
• Liposomal components may cause an allergic response.
• The potential for dumping as a result of poor management.
• Quick removal from circulation as a result of absorption.
Application:
Drug administration and health condition management are two areas in which lipomers, or lipid-polymer hybrid particles, are employed.
1.Liposomes for the respiratory system. Liposomes were are generally useful for addressing a wide range of lung disorders. Liposomal pressure enables the structure of goods in cans to be more accommodating, less disruptive, less harsh, and soft. The 2023 edition of Biological Forum: An International Journal, volume 15, issue 5, pages 33–38 Kardile as well as colleagues 36. When designing liposomes over pulmonary delivery, several issues must be considered, including drug/lipid proportion, composition, estimation, expenditure, and manner of conveyance. Depending on the nebulization method, the inhalation form may be dry or liquid. Liposomes made from powdery medicines require techniques such as spray drying or refining.
2.Liposomes within Ophthalmic diseases: Recently authorized liposomal formulations for the treatment of eye diseases include endophthalmitis, proliferative vitreoretinopathy, eye irritation, keratitis, failed corneal implants, as well as uveitis retinopathy.
3.Liposomes vaccine adjuvants: have been shown to increase both cell- and non-cell mediated immunity, making them a suitable vaccine adjuvant therapy. Phosphatidyl serine allows liposomes to be more precisely targeted for assembly on lymphoid cells. Liposomal immunization includes the cytokinesis of soluble antigens, vaccination-eligible organisms, as well as damaged deoxyribonucleic acid.
4.Liposomes for Brain Targeting: Because liposomes are biodegradable and harmless, they are utilized in brain sedative delivery systems. Connected to brain drug transport vectors, unilamellar vesicles (SUVs) can traverse the blood-brain barrier (BBB) by transcytosis or absorption. Stacking medications can more readily cross the BBB when encased in mannose-coated liposomes, which penetrate the brain. The brain-blood-barrier (BBB) is more easily crossed by mannose-coated liposomes, which enable stacked medications to penetrate into the brain. The neuropeptides met enkephalin dynorphin and leuenkephaline generally do not cross the blood-brain barrier when given directly. This strategy is very flexible, which is why the antidepressant amitriptyline often crosses the blood-brain barrier.
5.Liposome as Anti-Infective Agent: By concentrating the medication using a liposomal carrier, diseases such as tuberculosis, aspergillosis, erythrocytosis, histoplasmosis, candidiasis, and giardiasis can all be treated.
6.Liposome in Cancer Therapy: All cancer drugs have definite side effects over the long run. With fewer negative side effects, the liposomal approach directs the drug toward the growth. The tiny, stable liposomes are latently targeted toward different tumors because of their long-range mobility.
7.Liposomes in Cosmetics: Liposomes are used in the cosmetics business because they are physiochemically comparable to the cell membrane and have the potential to release chemicals into cells. 8.Liposomes in Intracellular Drug Delivery: Because liposomes can store higher drug concentrations than extracellular fluid, liposomal delivery of medications that typically enter the cells by pinocytosis can be particularly effective. Certain medications, often not well absorbed into cells, can have their cytosolic delivery increased with liposomes.
9. Liposomes in Sustain Release Drug Distribution: When taking medication that requires a sustained release mechanism for achieving and keeping a therapeutically adequate concentration, it is usual that one needs to take it many times per day.
10. Liposomes in Gene Therapy: In recent years, several attempts have been undertaken to sustain gene activity by delivering the proper foreign genes or DNA into cells.
Main Components of Liposomes:
Table 2 shows that the main components used in the commercialized products are glycerol phospholipid (GP), sphingomyelin (SM), and cholesterol. In GP, glycerol bridges two hydrophobic fatty acid chains and a hydrophilic polar head group (E, 2015).Figure 3 a depicts the different types of polar heads and fatty acids. Various head categories generate liposomes with neutral (PC and PE) or negative (PA, PS, PG, and cardiolipin) charges at physiological pH (A.G., 2014).
|
Product Name
|
Size
|
Structure
|
Main Composition
|
|
Doxil/Caelyx
|
ca. 100 nm
|
SUVs
|
HSPC, MPEG-DSPE, Chol.
|
|
AmBisome
|
50–100 nm [13]
|
SUVs
|
HSPC, DSPG, Chol
|
|
DaunoXome
|
45–80 nm [14]
|
SUVs
|
DSPC, Chol.
|
|
Marqibo
|
130–150 nm
|
SUVs
|
SM, Chol
|
|
Onivyde
|
ca. 110 nm
|
SUVs
|
DSPC, MPEG2000-DSPE, Chol.
|
|
Visudyne
|
<100 nm
|
SUVs [15]
|
EPC, DMPC
|
|
Shingrix
|
50–100 nm [16]
|
SUVs
|
DOPC, Chol.
|
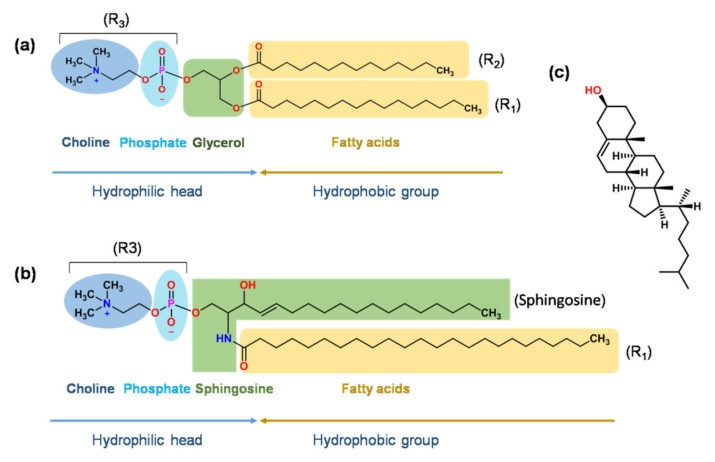
Figure 3:
a) Illustration of the structure of glycerol phospholipid. The fatty acids found in R1 and R2 represent unsaturated as well as saturated fats, which also comprise lauric, palmitic, oleic, myristic, stearic, and erucic acid. R3 might cardiolipin, phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, phosphatidic acid, include phosphatidyl inositols, and phosphatidylglycerol;
(b) The sphingomyelin a framework.
c) The chemical composition of cholesterol.
The DSPG in Vyxeos prevents liposomes coming from clumping collectively by using a strong Coulombic repulsive force, but the DSPG in Ambisome (an injection-ready ambisome liposome) may form a stable ionic complex with AmpB's positively charged amine group (Y., 2020) (Vyxeos, 2021). Vyxeos, Onivyde, and Daunoxome are lipid injections containing DSPC, a neutral synthetic lipid with a high phase transition temperature (Tm of 55 °C), well-defined fatty acid composition (two molecules of stearic acid), and high purity. Vereporfin powder is included in the infusion solutions Myocet and Visudyne, which also contain EPC as an excipient. The egg yolk (NPL) is a natural source of the phospholipid EPC. NPL has a wide transition temperature, making it challenging to generate identical NPL within liposomes, because might vary from batch to batch (Y., 2020) . Yet it costs less to produce than semi-synthetic and synthetic lipids. Furthermore, EPC's unsaturated fatty acid generates a low phase transition temperature of 15~-5 °C (J., 2015), indicating that the drug is "leaky" along with the liposome bilayer disordered at ambient temperature. Myocet, which contains EPC, is unstable in blood since the majority of medicines disintegrate within 24 hours (R., 2017) When serum is present, visudyne containing DMPC as well as EPC became less durable. Because of the prevalence of low-density lipoprotein (LDL) receptors in neovasculature, verteporfin moves quickly from the damaged liposome membrane to plasma lipoproteins, where it binds and reaches high concentrations (P., 2013) (Visudyne., 2021) . The covalent connection between MethoxyPEG (Mw 2000 Da) and DSPE (MPEG-DSPE) enables the creation of the "stealth" and sterically stable liposomes seen in Onivyde and Doxil. Both the mole percentage of PEG-DSPE in the lipid composition and the mole weight of PEG influence bilayer packing, circulation duration, as well as thermodynamic stability. Liposomes can prevent binding serum proteins as well as additional clearance through the mononuclear phagocytic system (MPS) by grafting a high molecular weight polyethylene glycol (PEG) molecule (>2000 Da) onto the lipid headgroup, while also reducing interaction as well as endocytosis of these liposomes by targeted cells (S., 2020) PEG's low molecular weight (>750 Da) results in limited steric stabilization (O., 2005). Additionally, a typical proportion of 5 mol% of PEG-lipid conjugates in live organisms (such as Doxil) create liposomes having the maximum biological stability when the concentration of PEG-DSPE in lipid assemblies is 7 ± 2 mol% (O., 2005) (.Y., 2002) . When the PEG-DSPE concentration is less than 4 mol%, the PEG chains create a "mushroom" shape and are around 3.5 nm thick.At concentrations ranging from 4 to 8 mol%, the PEG chain forms a "brush" having a thickness of 4.5 to 10 nm (O., 2005) (Z., 2012) . Raising the molar ratio further generates micelles rather than liposomes. All three of these medications— Exparel, DepoCyte, and DepoDur—have comparable lipid elements but distinct architectures. To generate MVLs, the formulation must contain an amphipathic lipid and a neutral lipid, such as vegetable oil, triglycerides, as well diglycerides (S., 1998) . Amphipathic zwitterionic phospholipids, notably DEPC as well as DOPC, produce the chambers' honeycomb-like walls. The negative charge of DPPG allows MVLs to avoid aggregation (Depodur., 2021) . Neutral lipids, such as triglycerides as well as triolein, act as hydrophobic gap fillers at bilayer intersection locations, preserving membrane connections (S., 2002) . The concentration of neutral lipids throughout the formulation impacts the capture volume and encapsulation efficiency of MVLs (S., 2002) . GPs are essential for formulations because they affect the biophysical features of liposomes (drug encapsulation, stability, and release) as well as its pharmacokinetics as well as pharmacodynamics actions towards live beings (W., 2017). The length, symmetry, inter- and intramolecular connections, branching, along with degree of unsaturation of the hydrocarbon chains all influence the bilayer's thickness and fluidity, as well as the phase transition temperature and drug release rate (A.G., 2014) (H., 1979) . Defined, a longer hydrocarbon chain can result in tighter membrane packed and greater drug retention, while a higher unsaturation or branching degree might result in looser membrane encapsulation. This is most likely due to cholesterol's preferential interaction with saturated phospholipids over unsaturated ones (S., 1995) (Y, 2016) .Sphingomyelin (SM) possesses a structure similar to a glycerol phospholipid (Figure 2b), but it contains sphingosine rather than glycerol (R., 2005) . Marqibo (vincristine sulfate liposome injection) dramatically reduces lipid hydrolysis within acidic environments and improves liposome durability by forming a bilayer membrane regarding SM. A transmembrane gradient of pH is commonly used to load active medicines in an acidic environment (pH 2.0-4.0). At 37 °C and pH 2.0, the hydrolysis rate of DSPC/Chol liposomes was about 100 times slower than that of SM/Chol liposomes (55/45, mol/mol) (S., 1998). Furthermore, the pharmacokinetic parameters of liposomes carrying SM/Chol were ideal, with longer circulation times and improved drug distribution to target tissues (A., 2013) . Cholesterol (Chol) is an additional structural component of the liposome bilayer (Figure 2c) that is found in nearly all commercial goods (Table 2). Incorporating Chol may enhance bilayer formation and lipid chain packing (S., 2018) , regulate membrane fluidity as well as stiffness (A., 2021) , and influence drug release (N., 2018) (M., 2017) , liposome stability (C., 1980), and exocytosis kinetics (N., 2016) Chol, at a 2:1 (Chol: QS21, w/w) ratio, can prevent the hydrolysis of QS21, an immune enhancer in the AS01B liposomal adjuvant system, which also comprises the Shingrix herpes zoster vaccine and the glycoprotein E antigen (N.M.C., 2005) . When comparing the AmBisome product's liposomal formulation to its non-sterol version, cholesterol lessens the former's toxicity (A.M, 1997). Chol has a concentration-dependent influence on bilayer characteristics. The characteristics of the lipid bilayer did not alter significantly at either the low (2.5 mol %) or large (>30 mol %) Chol dosages (S., 2018). Chol's "condensing influence" or "ordering effect"caused a progressive rise in particle size from 220 nm to 472 nm, decreased membrane fluidity, as well as reduced drug release at concentrations of 5 < mol% < 30. There have been studies on Chol and other sterols with similar structures, such as progesterone, ergosterol, and lanosterol, and how they affect membrane stiffness as well as stability (M., 2017) (R., 2015).
Liposome Preparation:
1. Film-Hydration Method:
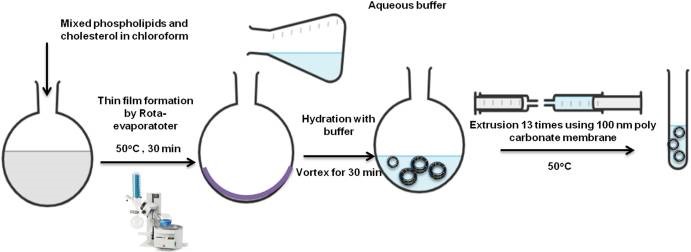
This procedure requires dissolve the hydrophobic medication and all lipids in a suitable organic solvent using a round-bottomed flask (E., 2021). The organic solution was then gradually evaporated at low pressure, leaving a thin film layer (A., 2013). We then use an aqueous buffer solutions to hydrate the resulting thin film when the temperature is higher than the lipid's transition temperature (Tm). The hydration solution may contain a hydrophilic drug or drugs in order to implant them inside the liposomes' watery its core. A more gradual rate of hydration results in higher encapsulation efficiency (E., 2021), with drug encapsulation efficiency determined by its rate of hydration. Liposome resizing, lamellarity types, and particle distributions may all be managed using bath or probe sonicators, or by extruding via polycarbonate membranes that have a particular pore diameter. Extrusion produces liposomes that are more stable and have a higher encapsulation efficiency than SUV liposomes, which may undergo hydrolysis or destruction of the containing lipids and/or medications. Metals contamination is possible while testing liposome liquids using probe sonication (Figure 4). (Z., 2012).
2.Reverse-phase evaporation method:
Organic solvents and sonication conditions have the ability denature therapeutic properties. Peptides that although this method is effective for molecules having a high molecular weight (F., 1980). One typical replacement for thin-film hydration is the reverse-phase evaporation process, which produces a water-in-oil emulsion (P., 2015) . Another aqueous buffer containing the hydrophilic medicine is instantly mixed with lipids within an organic solvent. Using a reduced pressure rotary evaporator, lipid vesicles develop and then dispersed in the water solution when the organic solvent evaporates. The extrusion is an effective method for reducing polydispersity along with the average size of generated vesicles (N.Q., 2017) .
3.Double-Emulsification Method:
To create MVLs, three commercial products from DepoCyte, DepoDur, and Expel employ this approach, also known as DepofoamplatformTM. Typically, the entire manufacturing process consists of four successive operations:
a)Production of a "water-in-oil" emulsion;
b)Formation of a "water-in-oil-in-water" emulsion.
c)Solvent extraction using stripping gas or vacuum pressure, and
d)Microfiltration for drug removal, concentration, and solution exchange (Q., 2000) (S., 2002).
Aseptic assurance is required throughout the manufacturing process for MVLs due to their small particle size, which cannot be produced as sterile batches utilizing 0.22 µm filtering. Lu et al. (B., 2021) . Investigated the effect of the procedure on critical quality features of bupivacaine MVLs and discovered that the particle size of the initial emulsion rises with increasing lipid content, and shearing speed has a significant influence on particle size. Because some MVLs collapse and medicine leaks into the interior aqueous phase during the solvent removal process, the encapsulation efficacy of the second emulsion decreases. Furthermore, high temperatures cause lipid bilayers to shift and restructure, resulting in lipid fusion and the collapse of the aqueous chambers.
4.Solvent Injection Methods:
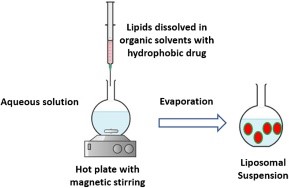
The injection processes were classified into different groups based on the organic solvents that were used (Figure 5) (C., 2020) . The hydrophobic active chemicals and lipids were dissolved in an organic solvent before being swiftly transferred to an aqueous phase. Throughout the mixing procedure, diethyl ether allows for direct solvent evaporation at temperatures higher than the solvent's boiling point (F., 1978). To inject ethanol, you must first create an aqueous solution that is ten to twenty times bigger than the original ethanol. The resulting solution can be vacuum evaporate using a rotary evaporator, dialysis, or filtration. Using this strategy, the majority of liposomal formulations with higher polydispersity indices (PDI) were created (C., 2011) . To make matters worse, prolonged exposure to high temperatures and organic solvents may impair the long-term integrity of medicines including lipids. (B., 2011).
5. Microfluidic Channel Method:
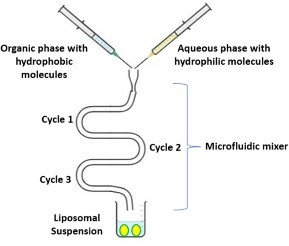
Figure 6.
Depicts the findings of a novel technique to liposome manufacturing that employs microfluidic tubes. Microfluidics enables the use of liquids in extremely narrow channels (B., 2009) This approach involves dissolving lipids in ethanol or as isopropanol, then injecting them vertically or counter-currently through micro-channels holding the water-based medium. This approach produces liposomes by continuous axial mixing of organic and aqueous solutions. Surfactants stabilize liposomes, preventing them from clotting and separating (D., 2016) . Microfluidic channel methods manage the process of merging organic and aqueous phases into liposomes that are consistently sized, polydisperse, morphologically as well as lamellar (L., 2021) .
Evaluation Of Liposomes:
Liposomes prepared using a variety of processes should have their physical, chemical, and biological characteristics assessed since these affect how the liposomes behave in living things.
Mathematical Aspects:
Size of particles the following technique can be used to ascertain these.
a) Dispersion of laser light.
b) Electron microscopy by transmission.
Surface charge:
The head groups' composition is what gives the liposomes their passive, negative, or natural charge. The liposomes' surface charge controls their interaction with the 19 target cells as well as the speed and extent of their dissemination in vivo. The surface charge measurement technique is based on the free-flow electrophoresis of MLVs. It makes use of a cellulose ester plate that has been immersed in pH 8.8 sodium borate buffer.After applying several moles of lipid samples to the plate, it is electrophoresed at 4°C for thirty minutes. Based on the charge on their surface, the liposomes split into two halves. Percentage of medicine included the amount of medication included in the liposomes aids in estimating how the medication will behave in a biological system. To calculate the percentage of drug encapsulation, the free drug fraction and the encapsulated drug fraction must first be separated. Next, employing the appropriate detergents, the encapsulated drug fraction is produced to be liposome-free in an aqueous solution. The following techniques aim to extract the free drug from the sample.
a) Protamine aggregates method.
b) Mini column centrifugation method Phase conduct:
c)Temperature liposomes experience a reversible phase transition at the transition. The TC serves as a marker for both the drug entrapment zone and stable permeability. DSC is in charge of it.
• Rate Of Drug Release:
In vivo assays, which aid in the prediction of the drug's pharmacokinetics and bioavailability, can be used to measure the rate at which the drug releases from the liposomes. On the other hand, in vivo research is thought to be more thorough. For the investigation, liposomes containing the tracer insulin are used. This insulin is favored because it is exclusively produced inside the ECF and rapidly excretes the facial tracer through the kidneys in conjunction with the liposomes' release of the tracer, which degrades at aconsistent pace. Determining the chemical properties of phospholipid: Assays such as the Bartlett and Steward assays are frequently used to directly assess the amount of phospholipid present in liposomes. The Bartlett Assay Because of its high sensitivity, this phospholipid detection method should detect even minute levels of phosphate incorrectly. Thus, double-water and borosilicate glass tubes are used.
1. The sample's lipid bilayer's phosphorous is first hydrolyzed to produce inorganic phosphate.
2. Next, in order to change inorganic phosphate into phosphomolybdic acid (PMP), ammonium molybdate is added.
3. Then, amino naphthyl sulphonic acid is added to the sample to quantitatively reduce the PMP into a blue-colored molecules.
4. To determine the amount of phospholipids present, the intensity of the blue color produced can be determined using spectrophotometric methods. The value obtained is displayed on a standard curve.
a) The Steward Assay: The shortcomings of the Bartlett test are addressed by this assay; nonetheless, it is not applicable to a mixture of unidentified phospholipids.
1.Using a 0.1 M solution of ammonium ferro-thiocyanate reagent and a known concentration of phospholipids in chloroform, the standard curve is created.
2.The same reagent is applied to the sample as well, and the optical density at 485 nm is measured.
3.The concentration of phospholipids can be determined by plotting the sample's absorbance on the standard curve.
a) The analysis of cholesterol:
The qualitative study was conducted using a capillary column that was packed full of fused silica.
c) Analysis using numbers: An interaction between the sample and a reagent causes the purple color complex to absorb light at 610 nm, which is.
Biological Description:
Infertility Anaerobic or aerobic cultures are used to perform the sterility test. The pyrogenicity of The Limulus amebocyte lysate (LAL) test is used to perform the pyrogenicity test. Toxicity to animals In order to conduct an animal toxicity test, histology, pathology, and survival rate are tracked (K., n.d.) (S., n.d.).
CONCLUSION:
In summary, liposomes are versatile drug carriers, particularly for focused drug delivery. Because of their structural makeup, liposomes are an adaptable nano-carrier that can be used to load and deliver a variety of medications or moieties for different disease situations. The creation of a stable, effective liposomal medication formulation requires an understanding of the significance of each structural element in the liposome formation process. To optimize the drug development process, prospective studies on the biocompatibility, circulation rate, and toxicity parameters of possible drug loaded liposome formulations must be conducted.Furthermore, there is a great deal of potential for this class of drugs to be taken into consideration as a preferred drug delivery strategy because the introduction of combination drug therapy and the development of novel drug products using liposomal formulations have demonstrated significant advantages over conventional drug therapies.
REFRENCES
-
-
-
- ABBOUD R., GREIGE-GERGES H., CHARCOSSET C.(2015) Effect of Progesterone, Its Hydroxylated and Methylated Derivatives, and Dydrogesterone on Lipid Bilayer Membranes. J. Membrane Biol. ;248:811–824. doi: 10.1007/s00232-015-9803-z. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- AKBARADEH A., REZAEI-SADABADY R., DAVARAN S. ,JOO S.W.,ZARGHAMI N.,HANIFEHPOUR Y.,SAMIEI M., KOUHI M., NEJATI-KOSHKI K.(2013) Liposome: classification, preparation, and applications. Nanoscale Res. Lett. ;8:102. [PMC free article] [PubMed] [Google Scholar] [Ref list]
- ALLARA, D.L.(2005) A perspective on surfaces and interfaces. Nature 437, 638–639. [Google Scholar] [CrossRef]
- ALLARA, D.L.; NUZZO R.G. (1985) Spontaneously Organized Molecular Assemblies. 1. Formation, Dynamics, and Physical Properties of n-alkanoic acids adsorbed from solution on an oxidized aluminum surface. Langmuir 1, 45–52. [Google Scholar] [CrossRef]
- ALYING C.R., BECK Z., MATYAS G. R., RAO M.(2016) Liposomal adjuvants for human vaccines. Expert Opin. Drug Deliv.13:807–816. doi: 10.1517/17425247.2016.1151871. [DOI] [PubMed] [Google Scholar]
- AMARNATH S, SHARMA US.(1997) Liposomes in drug delivery: progress and limitations. Int J Pharm. 154:123–140.
- BAYDA S.; ADEEL, M.; TUCCINARDI, T.; CORDANI, M.; BIZZOLIO, F.(2020) The history of nanoscience and nanotechnology: From chemicalphysical applications to nanomedicine. Molecules 25, 112. [CrossRef]
- BEIRANVAND S., EATEMADI A., KARIMI A. (2016) New Updates Pertaining to Drug Delivery of Local Anesthetics in Particular Bupivacaine Using Lipid Nanoparticles. Nanoscale Res. Lett.11:307–317. doi: 10.1186/s11671-016-1520-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BHATTARAI A., LIKOS E.M., WEYMAN C.M., SHUKLA G.C. (2021) Regulation of cholesterol biosynthesis and lipid metabolism: A microRNA management perspective. Steroids. 173:108878. doi: 10.1016/j.steroids.2021.108878. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- BHUPENDRA P., NARENDRA K., SUMAN S., AMIT R. (2015) Liposome: method of preparation, advantages, evaluation and its application. J. Appl. Pharmaceut. Res. 3:1–8. [Google Scholar] [Ref list]
- BOROCHOV H., SHINITZKY M., BARENHOLZ Y. (1979) Sphingomyelin phase transition in the sheep erythrocyte membrane. Cell Biochem. Biophys. 1:219–228. doi: 10.1007/BF02783664. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- CARUGO D., BOTTARO E., OWEN J., STRIDE E., NASTRUZZI C.(2016) Liposome production by microfluidics: potential and limiting factors. Sci. Rep. 6:25876. [PMC free article] [PubMed] [Google Scholar] [Ref list]
- DEPODUR. [(accessed on 20 June 2021)]; Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=Basic Search. Process [Ref list]
- GARBUZENKO O., BARENHOLZ Y., PRIEV A. (2005) Effect of grafted PEG on liposome size and on compressibility and packing of lipid bilayer. Chem. Phys. Lipids.135:117–129. doi: 10.1016/j.chemphyslip.2005.02.003. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- GARCON N.M.C., FRIEDE M. (2005) Vaccines Contraining a Saponin and a Sterol. US2005/0214322A1. U.S. Patent. September 29; [Ref list]
- GKIONIS L., AOJULA H., HARRIS L.K., TIRELLA A. (2021) Microfluidic-assisted fabrication of phosphatidylcholine-based liposomes for controlled drug delivery of chemotherapeutics. Int. J. Pharm. 604:120711. [PubMed] [Google Scholar] [Ref list]
- HAS C., SUNTHAR P. (2020) A comprehensive review on recent preparation techniques of liposomes. J. Liposome Res. 30:336–365. [PubMed] [Google Scholar] [Ref list]
- HILLERY A.M. (1997) Supramolecular lipidic drug delivery systems: From laboratory to clinic A review of the recently introduced commercial liposomal and lipid-based formulations of amphotericin B. Adv. Drug Delivery Rev. 24:345–363. doi: 10.1016/S0169-409X(96)00496-6. [DOI] [Google Scholar]
- HILLERY A.M. (1997) Supramolecular lipidic drug delivery systems: From laboratory to clinic A review of the recently introduced commercial liposomal and lipid-based formulations of amphotericin B. Adv. Drug Delivery Rev. 24:345–363. doi: 10.1016/S0169 409X(96)00496-6. [CrossRef] [Google Scholar] [Ref list]
- HYNONEN, U.; PALVA, A. (2013) Lactobacillus surface layer proteins: Structure, function and applications. Appl. Microbiol. Biotechnol. 97, 5225–5243. [Google Scholar] [CrossRef] [Green Ve rsion]
- JAAFAR-MAALEJ C., CHARCOSSET C., FESSI H. (2011) A new method for liposome preparation using a membrane contactor. J. Liposome Res. 21:213–220. [PubMed] [Google Scholar] [Ref list]
- JAIN NK. Controlled and novel drug delivery.
- KADDAH S., KHREICH N., KADDAH F., CHARCOSSET C., GREIGE-GERGES H. (2018) Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food Chem. Toxicol. 113:40–48. doi: 10.1016/j.fct.2018.01.017. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- KIM S., HOWELL S.B. (1998) Multivesicular Liposomes Having a Biologically Active Substance Encapsulated Therein in the Presence of a Hydrochloride. 5,723,147. U.S. Patent. 1998 March 3; [Ref list]
- KIRBY C., GREGORIADIS G. (1980) the effect of the cholesterol content of small unilamellar liposomes on the fate of their lipid components in vivo. Life Sci. 27:2223–2230. doi: 10.1016/0024-3205(80)90388-4. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- KOHLI A.G., KIERSTEAD P.H., VENDITTO V.J., WALSH C.L., SZOKA F.C. (2014) Designer lipids for drug delivery: From heads to tails. J. Control. Release. 190:274–287. doi: 10.1016/j.jconrel.2014.04.047. [PMC free article] [PubMed] [CrossRef] [Google Scholar] [Ref list]
- LARGE D.E., ABDELMESSIH R.G., FINK E.A., AUGUSTE D.T.(2021) Liposome composition in drug delivery design, synthesis, characterization, and clinical application. Adv. Drug Deliv. Rev. 176:113851. [PubMed] [Google Scholar] [Ref list]
- LI J., WANG X., ZHANG T., WANG C., HUANG Z., LUO X., DENG Y. (2015) A review on phospholipids and their main applications in drug delivery systems. Asian J. Pharm. Sci. 10:81–98. doi: 10.1016/j.ajps.2014.09.004. [CrossRef] [Google Scholar] [Ref list]
- LIU Y., MEI Z., MEI L., TANG J., YUAN W., SRINIVASAN S., ACKERMANN R., SCHWENDEMAN A.S.(2020) Analytical method development and comparability study for AmBisome® and generic Amphotericin B liposomal products. Eur. J. Pharm. Biopharm. 157:241–249. doi: 10.1016/j.ejpb.2020.09.008. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- LU B., MA Q., ZHANG J., LIU R., YUE Z., XU C., LI Z., LIN H. (2021) Preparation and characterization of bupivacaine multivesicular liposome: A QbD study about the effects of formulation and process on critical quality attributes. Int. J. Pharm.598:120335. doi: 10.1016/j.ijpharm.2021.120335. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- LU, C.; WANG, Y.; YANG, S.; WANG, C.; SUN, X.; LU, J.; YIN, H.; JIANG, W.; MENG, H.; RAO, F. (2018) Bioactive Self-Assembling Peptide Hydrogels Functionalized with Brain Derived Neurotrophic Factor and Nerve Growth Factor Mimicking Peptides Synergistically Promote Peripheral Nerve Regeneration. ACS Biomater. Sci. Eng. 4, 2994–3005. [Google Scholar] [CrossRef]
- LUO R., LI Y., HE M., ZHANG H., YUAN H., JOHNSON M., PALMISANO M., ZHOU S., SUN D. (2017) Distinct biodistribution of doxorubicin and the altered dispositions mediated by different liposomal formulations. Int. J. Pharm. 519:1–10. doi: 10.1016/j.ijpharm.2017.01.002. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- MAHERANI B., ARAB-TEHRANY E., MOZAFARI M., GAIANI C., M L. (2011) Liposomes: a review of manufacturing techniques and targeting strategies. Curr. Nanosci. 7:436 452. [Google Scholar] [Ref list]
- MANSOORI A. (2012) A review on liposomes. IJARPB. 2(4):453–464.
- MANTRIPRAGADA S. (2002) a lipid based depot (DepoFoam® technology) for sustained release drug delivery. Prog. Lipid Res. 41:392–406. doi: 10.1016/S0163-7827(02)00004 8. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- MESSNER, P.; PUM, D.; SARA, M.; STETTER, K.O.; SLEYTR, U.B. (1986) Ultrastructure of the cell envelope of the archaebacteria Thermoproteus tenax and Thermoproteus neutrophilus. J. Bacteriol. 166, 1046–1054. [Google Scholar] [CrossRef] [Green Version]
- NAJAFINOBAR N., MELLANDER L.J., KURCZY M.E., DUNEVALL J., ANGERER T.B., FLETCHER J.S., CANS A. (2016) Cholesterol Alters the Dynamics of Release in Protein Independent Cell Models for Exocytosis. Sci. Rep. 6:33702–33712. doi: 10.1038/srep33702. [PMC free article] [PubMed] [CrossRef] [Google Scholar] [Ref list]
- NOGUEIRA E., GOMES A.C., PRETO A., CAVACO-PAULO A. (2015) Design of liposomal formulations for cell targeting. Colloids Surf. B. 136:514–526. doi: 10.1016/j.colsurfb.2015.09.034. [PubMed] [CrossRef] [Google Scholar] [Ref list].
- PAJEWSKI R., DJEDOVIC N., HARDER E., FERDANI R., SCHLESINGER P.H., GOKEL G.W. (2005) Pore formation in and enlargement of phospholipid liposomes by synthetic models of ceramides and sphingomyelin. Bioorg. Med. Chem. 2005;13:29–37. doi: 10.1016/j.bmc.2004.10.001. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- PERKINS W., MALININ V., LI X., MILLER B., SEIDEL D., HOLZMANN P., SCHULZ H., HAHN M. (2017) System for Treating Pulmonary Infections. 9,566,234 B2. U.S. Patent. 2017 February 14; [Ref list]
- PUM, D.; NEUBAUER, A.; GYOERVARY, E.; SÁRA, M.; SLEYTR, U.B. (2008) S-layer proteins as basic biomolecular building blocks S-layer proteins as basic biomolecular building blocks. Adv. Packag. 100, 2–8. [Google Scholar]
- RANI S, HIREMATH R. Textbook of Industrial Pharmacy. And others, editor.
- RICHTER A.M., WATERFIELD E., JAIN A.K., CANAAN A.J., ALLISON B.A., LEVY J.G.(1993) Liposomal delivery of a photosensitizer, benzoporphyrin derivative monoacid ring A (BPD), to tumor tissue in a mouse tumor model. Photochem. Photobiol. 57:1000–1006. doi: 10.1111/j.1751-1097.1993.tb02962.x. [DOI] [PubMed] [Google Scholar]
- SADEGHI N., DECKERS R., OZBAKIR B., AKTHAR S., KOK R.J., LAMMERS T., STORM G. (2018) Influence of cholesterol inclusion on the doxorubicin release characteristics of lysolipid-based thermosensitive liposomes. Int. J. Pharm. 548:778–782. doi: 10.1016/j.ijpharm.2017.11.002. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- SARAF S., JAIN A., TIWARI A., VERMA A., PANDA P.K., JAIN S.K. (2020) Advances in liposomal drug delivery to cancer: An overview. J. Drug Deliv. Sci. Technol. 56:101549. doi: 10.1016/j.jddst.2020.101549. [CrossRef] [Google Scholar] [Ref list]
- SHI N.-Q., QI X.-R. (2017) In: Liposome-Based Drug Delivery Systems. Lu W.-L., Qi X.-R., editors. Springer Berlin Heidelberg; Berlin, Heidelberg: 2017. Preparation of drug liposomes by reverse-phase evaporation; pp. 1–10. [Google Scholar] [Ref list]
- SILVERMAN J.A., DEITCHER S.R. (2013) Marqibo® (vincristine sulfate liposome injection) improves the pharmacokinetics and pharmacodynamics of vincristine. Cancer Chemother. Pharmacol. 71:555–564. doi: 10.1007/s00280-012-2042-4. [PMC free article] [PubMed] [CrossRef] [Google Scholar] [Ref list]
- SKUPIN-MRUGALSKA P., PISKORZ J., GOSLINSKI T., MIELCAREK J., KONOPKA K., DU ZGU NE? N. (2013) Current status of liposomal porphyrinoid photosensitizers. Drug Discov. Today. 18:776–784. doi: 10.1016/j.drudis.2013.04.003. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- SLEYTR, U.B.; SCHUSTER, B.; EGELSEER, E.M.; PUM, D. (2014) S-layers: Principles and applications. FEMS Microbiol. Rev. 38, 823–864. [Google Scholar] [CrossRef] [Green Version]
- SONG L.Y., AHKONG Q.F., RONG Q., WANG Z., ANSELL S., HOPE M.J., MUI B. (2002) Characterization of the inhibitory effect of PEG-lipid conjugates on the intracellular delivery of plasmid and antisense DNA mediated by cationic lipid liposomes. Biochim. Biophys. Acta Biomembr. 1558:1–13. doi: 10.1016/S0005-2736(01)00399-6. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- SZOKA F., JR., PAPAHADJOPOULOS D. (1980) Comparative properties and methods of preparation of lipid vesicles (liposomes) Annu. Rev. Biophys. Bioeng. 9:467 508. [PubMed] [Google Scholar] [Ref list]
- SZOKA F., JR., PAPAHADJOPOULOS D. (1978) Procedure for preparation of liposomes with large internal aqueous space and high capture by reverse-phase evaporation. Proc. Natl. Acad. Sci. U. S. A. 75:4194–4198. [PMC free article] [PubMed] [Google Scholar] [Ref list]
- TAKECHI-HARAYA Y., MATSUOKA M., IMAI H., IZUTSU K., SAKAI-KATO K. (2020) Detection of material-derived differences in the stiffness of egg yolk phosphatidylcholine-containing liposomes using atomic force microscopy. Chem. Phys. Lipids.233:104992. doi: 10.1016/j.chemphyslip.2020.104992. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- TAKECHI-HARAYA Y., SAKAI-KATO K., ABE Y., KAWANISHI T., OKUDA H., GODA Y. (2016) Atomic Force Microscopic Analysis of the Effect of Lipid Composition on Liposome Membrane Rigidity. Langmuir. 32:6074–6082. doi: 10.1021/acs.langmuir.6b00741. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- VARGA Z., WACHA A., VAINIO U., GUMMEL J., BO TA A. (2012) Characterization of the PEG layer of sterically stabilized liposomes: A SAXS study. Chem. Phys. Lipids. 165:387–392. doi: 10.1016/j.chemphyslip.2011.12.011. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- VEMURI S., RHODES C.T. (1995) Preparation and characterization of liposomes as therapeutic delivery systems: A review. Pharm. Acta Helv. 70:95–111. doi: 10.1016/0031 6865(95)00010-7. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- VISUDYNE. [(accessed on 20 June 2021)]. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/visudyne [Ref list]
- VYXEOS LIPOSOMAL (Previously Known as Vyxeos) [(accessed on 20 June 2021)]. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/vyxeos liposomal [Ref list]
- WANG M., LIU M., XIE T., ZHANG B., GAO X. (2017) Chitosan-modified cholesterol-free liposomes for improving the oral bioavailability of progesterone. Colloids Surf. B. 159:580–585. doi: 10.1016/j.colsurfb.2017.08.028. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- WEBB M.S., BALLY M.B., MAYER L.D., MILLER J.J., TARDI P.G. (1998) Sphingosomes for Enhanced Drug Delivery. 5,741,516. U.S. Patent. 1998 April 21; [Ref list]
- YE Q., ASHERMAN J., STEVENSON M., BROWNSON E., KATRE N.V. (2000) DepoFoam™ technology: A vehicle for controlled delivery of protein and peptide drugs. J. Control. Release. 64:155–166. doi: 10.1016/S0168-3659(99)00146-7. [PubMed] [CrossRef] [Google Scholar] [Ref list]
- YU B., LEE R.J., LEE L.J. (2009) Microfluidic methods for production of liposomes. Methods Enzymol. 465:129–141. [PMC free article] [PubMed] [Google Scholar] [Ref list]


 Shravani Gaikwad*
Shravani Gaikwad*
 Pranjal Chougule
Pranjal Chougule






 10.5281/zenodo.15050599
10.5281/zenodo.15050599