One of the most important issues in regenerative medicine has always been bone regeneration. Despite the substantial therapeutic benefits of scaffold-based and conventional bone grafting, these methods are frequently constrained by immunological reactions, infection risk, poor mechanical compatibility, and insufficient vascularization. Researchers have looked to nano-scaffold-based systems—an advanced class of biomaterials designed at the nanoscale to improve cellular interactions and replicate the natural extracellular matrix (ECM)—to get over these restrictions. To facilitate effective tissue regeneration, these nano-scaffolds combine molecular-level architecture, surface functionalization, and biological signaling. The most recent advancements in nano-scaffold technology for bone regeneration and repair are highlighted in this thorough analysis. In addition to surface-modified and bioactive coatings intended to enhance osteointegration and angiogenesis, it investigates a variety of nanostructured scaffolds, including polymeric, ceramic, metallic, and composite systems. The role of nanomaterials that improve mechanical strength and cellular performance—such as hydroxyapatite nanoparticles, carbon nanotubes, graphene oxide, and bioactive glasses—is given particular consideration. To assess the molecular pathways by which nano-scaffolds affect osteogenesis, vascularization, and immunological regulation, preclinical and clinical research is examined. Compared to conventional scaffolds, nano-scaffolds have a number of benefits, such as superior mechanical stability, controlled degradation, increased growth factor supply, and improved biomimicry. But there are still issues with long-term biocompatibility, large-scale production, and regulatory approval. Despite these challenges, continuous developments in biomaterials science and nanotechnology are driving nano-scaffolds toward practical clinical uses. According to the review's findings, nano-scaffold systems are an important advancement in regenerative medicine since they bridge the gap between biological functionality and structural support for successful bone healing.
Bone regeneration, Nano-scaffolds, Regenerative medicine, Nanotechnology, Biomaterials, Osteogenesis, Hydroxyapatite, Tissue engineering.
One of the human body's most intricate and active tissues, bone is able to continuously change and heal itself. It permits mobility, preserves mineral balance, protects important organs, and offers structural support. However, the body's ability to mend itself is frequently exceeded by critical-sized bone defects brought on by trauma, infection, tumor excision, or degenerative illnesses. In many situations, restoring both structure and function requires medical intervention.3-4
-
- Bone Biology and Healing Mechanism
Cellular proliferation, differentiation, and matrix formation are all part of the intricately coordinated biological process of bone repair. It goes through four main stages: remodeling, soft callus formation, hard callus formation, and inflammation. Following an injury, inflammatory cells like neutrophils and macrophages release signaling chemicals that attract mesenchymal stem cells (MSCs). While osteoclasts eliminate damaged tissue, these cells differentiate into osteoblasts, which release the extracellular matrix (ECM) and start the mineralization process. Over time, the extracellular matrix (ECM), which is mostly made up of collagen and non-collagenous proteins, mineralizes to create mature bone.
These actions are coordinated by growth factors such transforming growth factor-β (TGF-β), vascular endothelial growth factor (VEGF), and bone morphogenetic proteins (BMPs). Establishing an appropriate microenvironment that promotes both osteogenesis and angiogenesis is necessary for successful regeneration. Systems based on nano-scaffolds have been created expressly to mimic these microarchitectures and biological signals at the nanoscale.5
-
- Limitations of Conventional Scaffolds
For bone tissue engineering, conventional scaffolds made of polymers, ceramics, or composites have offered promising platforms. They act as three-dimensional structures that facilitate ECM deposition, cell adhesion, and proliferation. They often fail to replicate the nanoscale structure of actual bone tissue, though. Poor integration and mechanical mismatch can result from variations in pore size, surface roughness, and degradation kinetics.6
Furthermore, a lot of traditional scaffolds are biologically inert, lack bioactivity, and depend on protein surface adsorption for cell adhesion. This restricts their capacity to efficiently direct vascularization and cell differentiation. Dense scaffolds can also prevent vascular infiltration and nutrient diffusion, which can lead to necrosis or partial repair at the defect core. The development of nano-engineered scaffolds, which attempt to replicate the chemical and structural complexity of natural bone at the nanoscale, has been spurred by these constraints.7
-
- The Concept of Nano-Scaffolds
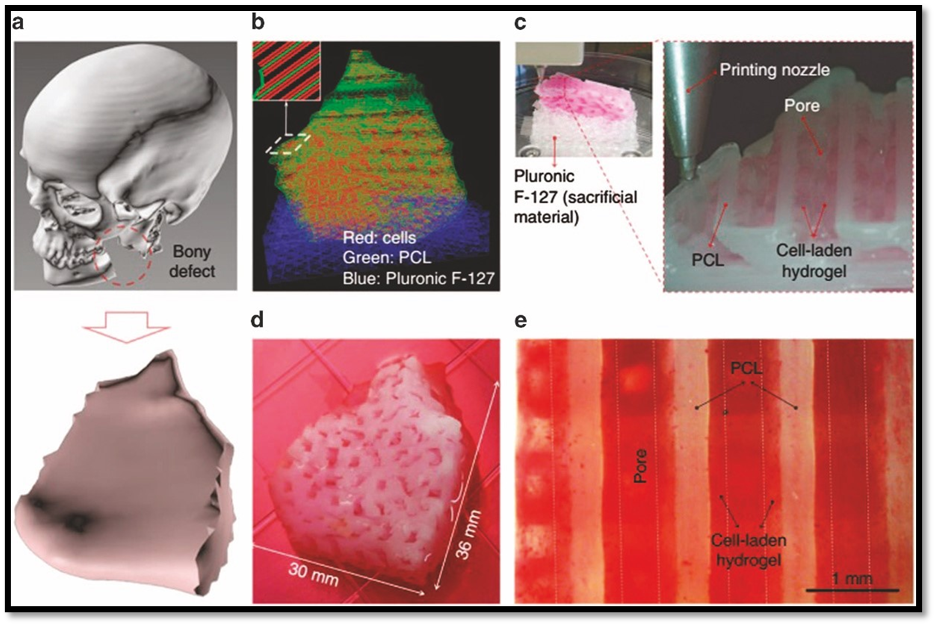
In order to more closely resemble the extracellular matrix, nano-scaffolds are sophisticated biomaterial systems that include nanostructured elements in their composition, surface modification, or architecture. These scaffolds' nanoscale topography increases osteogenic differentiation, integrin-mediated cell adhesion, and protein adsorption. Researchers can adjust biological signals, mechanical strength, and degradation rates by adding nanoparticles or nanofibers. Electrospinning, self-assembly, sol-gel processing, 3D printing, and phase separation are common methods for creating nano-scaffolds. These techniques allow for exact control over surface chemistry, pore size, and fiber diameter. For localized and long-term distribution at the defect location, nano-scaffolds can also be functionalized with growth factors, peptides, or medications. They are a great basis for intelligent bone regeneration systems because of their versatility.8-10
-
- Scope of the Review
A detailed overview of nano-scaffold-based approaches in regenerative medicine and bone tissue engineering is given in this article. The fundamentals of bone biology, the justification for nano-engineering, and the effectiveness of different nanostructured materials in preclinical and clinical settings are all covered. The article also examines the molecular interactions of nano-scaffolds with biological systems, as well as their manufacturing processes, difficulties, and potential applications in clinical settings.11-12
2. Bone Biology and Regeneration
As a living tissue, bone undergoes continuous remodeling due to a balance between creation and resorption. Its capacity for regeneration is dependent on molecular signaling, vascular supply, and cellular communication. Understanding the biology and structure of native bone is crucial to appreciating how nano-scaffolds aid in bone healing.13
2.1 Structure and Composition of Bone
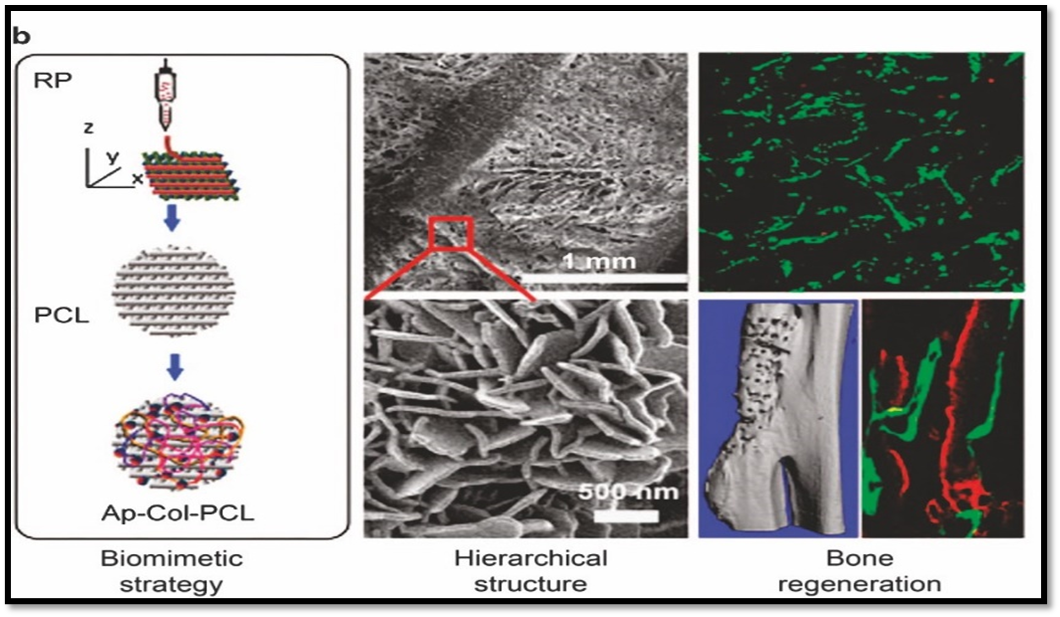
About 30% organic matrix and 70% inorganic material make up bone. Type I collagen and non-collagenous proteins, which offer flexibility and a scaffold for mineral deposition, make up the majority of the organic fraction. Hardness and compressive strength are imparted by the inorganic phase, primarily hydroxyapatite [Ca??(PO?)²(OH)?]. Bone is robust and resilient due to this mixed makeup. Bone tissue is divided into compact (cortical) and spongy (trabecular) areas at the microscopic level. While trabecular bone sustains marrow and circulatory networks, cortical bone offers strength and rigidity. The design of nano-scaffolds is heavily influenced by the hierarchical architecture of bone, which ranges from macroscopic structures to nanoscale collagen fibrils.14
2.2 Cellular and Molecular Mechanisms
Osteoblasts, osteoclasts, and osteocytes are the three main cell types involved in bone remodeling. Osteocytes function as mechanosensors that control the processes of osteoblasts creating new bone matrix and osteoclasts resorbing old bone. These cellular processes are coordinated by molecular pathways such TGF-β signaling, Wnt/β-catenin, and BMP. By offering nanoscale cues that affect adherence and gene expression, nano-scaffolds directly interact with these cells. Surfaces with nanoscale roughness, for example, promote osteoblast development and proliferation. In order to activate these biochemical pathways, certain nano-scaffolds are even designed to release growth factors or ions.15
2.3 The Role of Angiogenesis and Mechanical Support
The development of new blood vessels and the restoration of mechanical stability are essential for successful bone regeneration. To improve vascularization, angiogenic substances like VEGF or copper ions can be added to nano-scaffolds. Additionally, their nanostructured porosity facilitates nutrition transport and endothelial cell penetration. By mixing polymers and nanoparticles, nanocomposite scaffolds can minimize stress shielding and reach strength levels more akin to those of real bone.16
3. Nanomaterials Applied in Bone Repair and Regeneration
Materials that can restore both biological functionality and structural integrity are essential for bone regeneration. Nanomaterials have shown unmatched potential to imitate the natural extracellular matrix (ECM) at the molecular level among all biomaterials studied to far. Because of their size, which is usually between 1 and 100 nanometers, they can interact directly with biological molecules including proteins, enzymes, and cell receptors, improving tissue integration and cellular responses.17
The improved biological activity of nanostructures is combined with the mechanical support of traditional materials in nano-engineered scaffolds. These scaffolds offer particular surface topographies and chemical signals that control cellular adhesion, proliferation, and differentiation in addition to providing a framework for cell growth. The many kinds of nanomaterials used in bone regeneration and their modes of action are covered in the ensuing subsections.18
3.1 The Role of Nanotechnology in Bone Regeneration
Collagen fibrils (organic phase) and hydroxyapatite nanocrystals (inorganic phase) make up the nanocomposite substance that is natural bone. Scientists were motivated by this hierarchical structure to create synthetic scaffolds with comparable characteristics. Researchers can modify surface energy, roughness, and charge distribution—all of which affect protein adsorption and cellular behavior—by engineering materials at the nanoscale. It has been demonstrated that nanomaterials greatly improve both osteoinductivity (the capacity to promote new bone formation) and osteoconductivity (the capacity to facilitate bone growth along a surface). They enhance integration with host bone and hasten healing when utilized in scaffold construction. Nanomaterials are also appropriate for multifunctional regeneration systems because of their high surface area-to-volume ratio, which enables effective medication loading and regulated release of growth factors or antibiotics.19
In general, inorganic, polymeric, carbon-based, and composite nanomaterials can be employed for bone regeneration and repair. Different structural, mechanical, and biological traits are displayed by each category. 20
3.1.1 Nanomaterials that are inorganic
The most researched inorganic nanomaterials for bone applications are hydroxyapatite (HAp) and bioactive glass nanoparticles.
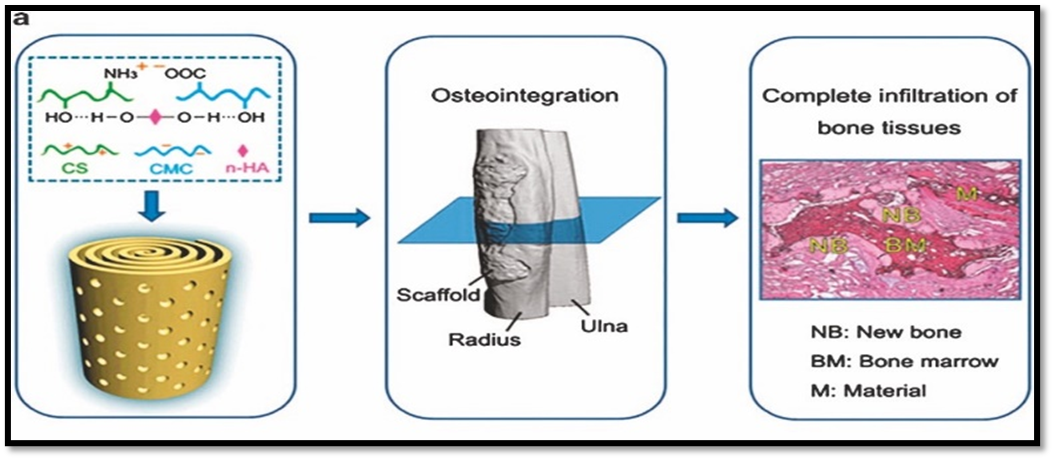
- Nano-hydroxyapatite (nHAp): Encourages osteointegration by imitating the mineral phase of bone. By increasing the bioactivity and surface roughness of scaffolds, nHAp particles promote osteoblast adhesion and proliferation.
- Glasses that are bioactive: These materials, which are mostly composed of SiO?, CaO, and P2O?, disintegrate slowly in physiological conditions and release ions that promote angiogenesis and osteogenesis. Excellent biocompatibility and biodegradability are provided by calcium phosphate nanoparticles. Local signaling is provided by their ion release (Ca²?, PO?³?), which speeds up mineralization. To increase mechanical strength and biological performance, inorganic nanoparticles are commonly added to polymeric or composite scaffolds.21
3.1.2 Polymeric Nanomaterials
Because they are biodegradable and may be tailored to manage mechanical strength and breakdown rates, both natural and synthetic polymers are useful building blocks for nano-scaffolds. Typical polymers consist of: Collagen, chitosan, gelatin, alginate, and silk fibroin are examples of natural polymers. These have built-in cell-binding motifs and are biocompatible. Polycaprolactone (PCL), polylactic acid (PLA), and polylactic-co-glycolic acid (PLGA) are synthetic polymers. To increase their osteogenic potential, they can be functionalized using nanoparticles. Electrospun polymeric nanofibers have drawn special interest. Their porosity structure and nanoscale diameter closely resemble the natural extracellular matrix (ECM), allowing for superior cell infiltration and nutrition exchange. Furthermore, polymeric nano-scaffolds are very useful for guided bone regeneration because they can encapsulate bioactive compounds for regulated release.21-25
3.1.3 Carbon-Based Nanomaterials
Because of their exceptional mechanical, electrical, and chemical characteristics, carbon nanomaterials like carbon nanotubes (CNTs), graphene oxide (GO), and carbon nanofibers (CNFs) have become attractive options for bone tissue engineering. CNTs, or carbon nanotubes: Boost electrical conductivity and tensile strength. By encouraging signal transduction via mechanosensitive pathways, they can promote osteogenic differentiation. Graphene oxide (GO): Provides oxygen-containing groups and a wide surface area that promote cell adhesion and protein adsorption. GO enhances the mechanical strength, mineral deposition, and biocompatibility of polymeric scaffolds. However, because their reactivity and form can occasionally cause oxidative stress in cells, carbon-based nanomaterials must be carefully tailored to avoid cytotoxicity.25-26
3.1.4 Metallic Nanomaterials
Implants are frequently coated with titanium dioxide (TiO?) nanoparticles to improve cell adherence and surface roughness. In physiological settings, magnesium nanoparticles progressively break down, releasing ions that support bone development while preserving local alkalinity to ward off infection. In order to avoid postoperative infections, silver (Ag) nanoparticles with potent antibacterial qualities are integrated into nano-scaffolds. These metallic nanoparticles are beneficial for therapeutic implants because they offer extra features including mechanical reinforcement and antibacterial activity.27
3.1.5 Composite Nanomaterials
Composite nano-scaffolds merge biological functioning with mechanical stability by combining two or more material types, usually polymers with metals or ceramics. For instance: Osteoconductivity and controlled degradation are enhanced by PLGA/nano-hydroxyapatite composites. Composites made of chitosan and graphene oxide improve cellular activity and strength. Elasticity and ion-releasing bioactivity are combined in silk fibroin–bioactive glass composites. Composite nano-scaffolds are very adaptable for bone regeneration applications because they enable customization of mechanical, chemical, and biological properties.28
3.2 Mechanisms of Action of Nanomaterials in Bone Healing
Nanomaterials' capacity to affect biological function at the molecular level gives them the possibility for regeneration: Protein adsorption: Serum proteins like fibronectin and vitronectin, which promote cell adhesion through integrin binding, are selectively adsorbed by nanostructured surfaces. Cell differentiation: Signaling pathways that support osteogenic gene expression, such as MAPK, Wnt, and BMP, are activated by nanoscale topography. Mineralization: By serving as calcium phosphate deposition nucleation sites, nanoparticles promote early mineralization. Angiogenesis: Endothelial cell migration and vascular development are stimulated by ion release from nanomaterials (such as Si?, Ca²?, and Cu²?). Antibacterial effects: By preventing bacterial adherence, metallic and oxide nanoparticles lower the risk of infection in bone defects. Instead of merely assisting regeneration, nano-scaffolds actively foster it by incorporating these biological processes.29
4. Influences on Bone Regeneration
A complex interaction of biological, chemical, and physical variables influences the highly coordinated process of successful bone regeneration. The degree to which these elements are tuned to mimic the natural bone microenvironment will determine how successful nano-scaffolds are in regenerative medicine. The rate and quality of bone production are determined by a number of factors, including scaffold surface chemistry and cellular signaling. Designing next-generation nano-scaffolds that can accomplish full and functional bone restoration requires an understanding of these impacts.30
4.1 Cellular and Molecular Interactions
The micro- and nanoscale environment of the scaffold substantially regulates the behavior of cells, which are the basic drivers of bone formation. Mesenchymal stem cells (MSCs), osteoblasts, and osteoclasts are extremely sensitive to the surface topography and chemical makeup of biomaterials. By offering nanoscale characteristics that mimic the natural extracellular matrix (ECM), nano-scaffolds affect these biological reactions. Cell adhesion, proliferation, and differentiation are all improved by this resemblance. Integrin binding is mediated by proteins like fibronectin and vitronectin adhering to the scaffold surface at the start of the interaction.31
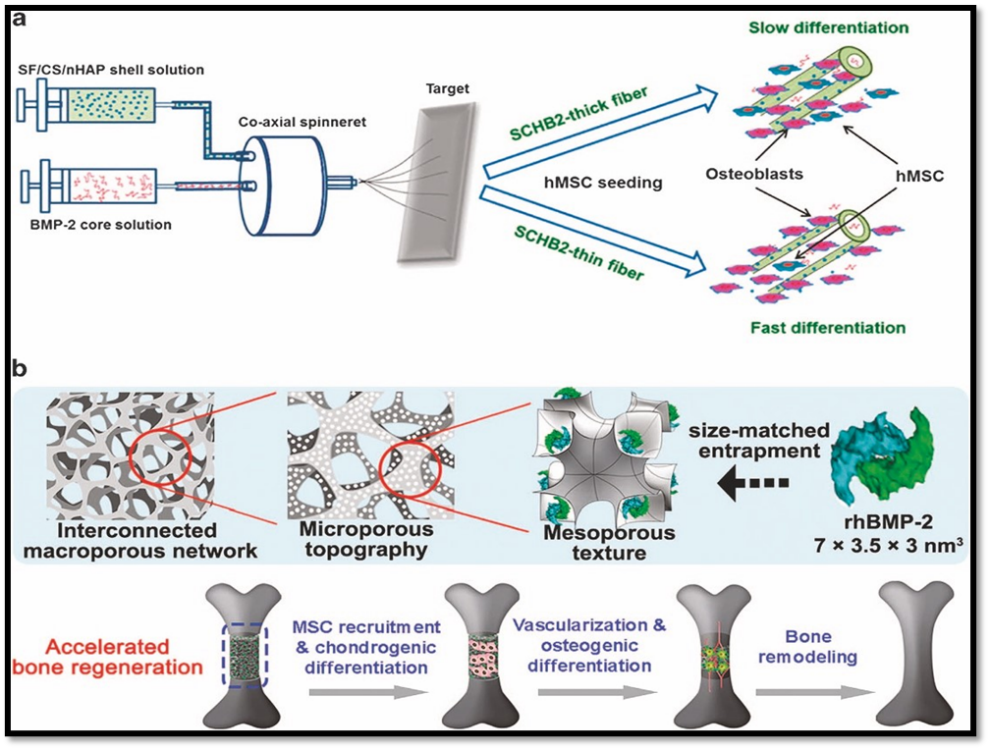
After adhering, cells detect the nanoscale signals and initiate intracellular pathways such as Wnt/β-catenin, BMP signaling, and MAPK, which eventually stimulate osteogenic differentiation. Moreover, bioactive compounds like growth factors, peptides, or cytokines can be added to nano-scaffolds to functionalize them and produce a favorable cellular microenvironment. Faster bone repair results from the controlled release of BMP-2, VEGF, and TGF-β from nanostructured carriers, which promotes angiogenesis and osteogenesis.32
4.2 Physical and Mechanical Factors
The response of bone tissue to mechanical stimulation is quite high. Cell destiny and tissue regeneration are significantly influenced by a scaffold's elasticity, porosity, and stiffness. The programmable structure of nano-scaffolds makes it possible to precisely tune these characteristics. Porosity and Pore Size: Sufficient porosity guarantees vascular infiltration, waste elimination, and nutrient diffusion. While interconnected macro-pores (>100 µm) promote capillary development and new tissue ingrowth, nanoscale pores (50–500 nm) boost protein adsorption and osteoblast adhesion. Surface Roughness and Stiffness: While ideal stiffness promotes mechanical stability, increased nanoscale roughness improves osteoblastic differentiation. Scaffolds that are too soft may collapse before tissue formation, whereas those that are too hard may produce stress shielding. Mechanical Loading: Cellular mechanotransduction pathways are impacted by dynamic mechanical stimuli as compression, shear, or vibration.33
These signals can be efficiently transmitted by nano-scaffolds made to resemble the mechanical characteristics of natural bone, encouraging matrix formation and mineralization. To sustain both biological activity and mechanical function during regeneration, these physical characteristics need to be carefully adjusted.34
4.3 Chemical and Surface
Bone regeneration is significantly impacted by the surface energy and chemical makeup of nano-scaffolds. Signaling cascades related to bone metabolism involve elements like calcium, phosphorus, silicon, zinc, and magnesium. These ions improve the osteoinductivity and angiogenic potential of nanomaterials.
For instance:
- Osteoblast development and mineralization are stimulated by calcium and phosphate ions.
- Silicon ions encourage vascularization and collagen formation.
In order to facilitate more effective cell attachment and proliferation, surface modification methods like plasma treatment, acid etching, or nanoparticle coating can further enhance surface wettability and protein adsorption. When it comes to producing bioactive interfaces, nano-scaffolds are preferable to traditional scaffolds because they can precisely control surface chemistry at the nanoscale.35-37
4.4 Biological Environment and Vascularization
One of the most important elements influencing the success of bone regeneration is vascularization. Rapid angiogenesis is the only way to provide the oxygen and nutrients that newly created bone needs. By releasing angiogenic factors like VEGF and offering topographical cues that direct endothelial cell migration, nano-scaffolds promote vascularization. It is common to refer to the interaction between angiogenesis (the production of blood vessels) and osteogenesis (the formation of bones) as a "coupled process." Better regeneration results are produced by nanomaterials that stimulate both at the same time. For instance, it has been demonstrated that bioactive glass nanoparticles doped with copper or cobalt ions promote osteogenic differentiation in MSCs and increase endothelial cell proliferation.38-40
4.5 Immune Response and Inflammation
Depending on the inflammatory response, the immune system can either aid or impede bone rebuilding. Chronic inflammation can result in fibrosis or implant failure, although a moderate, brief inflammatory phase is necessary to start tissue repair. By decreasing the release of pro-inflammatory cytokines and promoting macrophage polarization toward an M2 (pro-healing) phenotype, nano-scaffolds with biocompatible surfaces can alter immune cell activity. Titanium oxide or magnesium oxide nanoparticle surface coatings, for instance, have been shown to reduce immunological rejection while promoting osteogenesis. One of the new benefits of scaffolds based on nanomaterials is this immunomodulatory characteristic.41-42
4.6 Degradation and Ion Release Kinetics
Another important aspect affecting bone regeneration is the controlled breakdown of scaffolds. A scaffold should ideally break down gradually as new bone grows, preserving mechanical stability in the early stages of healing and enabling full resorption in the later stages. Because of their high surface reactivity, nano-engineered materials allow for fine control over degradation rates. The breakdown products of nano-scaffolds, especially bioactive ions, act as signaling molecules that encourage mineralization and cell differentiation. However, ion toxicity or local pH swings could result from unchecked or quick breakdown. For clinical applications, balancing degradation kinetics is still a crucial design factor.42-45
5. Bioactive Materials
One of the most important developments in the field of bone tissue engineering is the creation of bioactive materials. Bioactive materials are intended to actively interact with surrounding tissues, promoting cell adhesion, proliferation, and differentiation, in contrast to inert scaffolds that only function as mechanical supports. By releasing therapeutic ions or biomolecules directly at the defect site, these bioactive components are essential for promoting osteogenesis and speeding up bone healing in the context of nano-scaffolds.46
5.1 Concept of Bioactivity
The ability of a substance to cause a certain biological reaction at the interface between the substance and the host tissue is known as bioactivity. For bone regeneration, this usually entails activating osteogenic cells and depositing a layer that resembles hydroxyapatite to create a direct link with bone. Bioactive material-infused nano-scaffolds mimic the chemistry and structure of the extracellular matrix (ECM) seen in real bone. Faster ion exchange and protein adsorption are made possible by their huge surface area and nanoscale porosity, which improves integration with the host tissue. A scaffold's bioactivity is frequently assessed by how well it forms apatite in simulated bodily fluid (SBF) and how it affects osteoblast behavior.
5.2 Types of Bioactive Materials
Depending on their makeup and mode of action, bioactive materials employed in the creation of nano-scaffolds can be divided into a number of classes.47
5.2.1 Bioactive Glasses
Bioactive glasses have demonstrated remarkable osteoconductive and osteoinductive capabilities, especially those made of silicon dioxide (SiO?), calcium oxide (CaO), sodium oxide (NaOO), and phosphorus pentoxide (P2O?). These glasses produce ionic compounds that promote angiogenesis and osteogenic differentiation when submerged in bodily fluids. Nano-bioactive glass (nBG), the nanoscale variant, has various benefits. Increased surface area results in higher surface reactivity. Improved adhesion to hydroxyapatite and collagen Enhanced cell proliferation and quicker apatite formation in order to enhance mechanical strength and biological performance, nBGs are frequently added to polymeric scaffolds. It is known that their ionic dissolution products (Ca2+, Si2+, and P2+) upregulate osteogenesis-related genes such COL1A1, RUNX2, and ALP.
5.2.2 Bioactive Ceramics
The most popular materials for bone regeneration are bioactive ceramics including calcium silicate, tricalcium phosphate (TCP), and hydroxyapatite (HAp). Their composition is quite similar to bone's inorganic mineral phase. Improved bioresorption, increased osteoblast adhesion, and chemical resemblance to natural bone mineral are all demonstrated by nano-hydroxyapatite (nHAp). Antibiotics and growth hormones can also be transported by it. β-Tricalcium Phosphate (β-TCP): Osteoconductive and biodegradable, ideal for short-term scaffolds that progressively dissolve as bone grows again. Bioactive ions released by calcium silicate (CaSiO?) promote angiogenesis and osteogenesis. Bioactive ceramics create composite nano-scaffolds with superior mechanical stability and biofunctionality when mixed with polymers or other nanomaterials.
5.2.3 Polymer-Based Bioactive Composites
Because they include amino acid sequences that promote cell adhesion and migration, natural polymers like collagen, chitosan, gelatin, and silk fibroin are intrinsically bioactive. They frequently lack adequate mechanical strength, though. As a result, synthetic–natural hybrid composites are frequently employed.
For instance:
- Composites made of chitosan and nano-hydroxyapatite have both osteoconductivity and antibacterial activity.
- Collagen/bioactive glass scaffolds combine great bone-bonding capacity with outstanding biocompatibility.
- For load-bearing bone deformities, silk fibroin/calcium phosphate nanocomposites provide high flexibility and delayed deterioration. In order to improve long-term regeneration, these polymer-based composites can also be designed to release bioactive chemicals gradually.47-50
5.2.4 Metallic and Ion-Doped Bioactive Materials
When added in trace amounts, some metallic elements can greatly increase the bioactivity of scaffolds. Magnesium (Mg): Promotes the growth of bones and has a weak antimicrobial effect. Zinc (Zn): Encourages the production of collagen and alkaline phosphatase activity. Copper (Cu): Promotes tissue vascularization and angiogenesis. These metal ion-doped nano-scaffolds preserve regulated breakdown rates while enhancing cell survival and mechanical integrity. For instance, compared to undoped glass, magnesium-doped bioactive glass has demonstrated quicker bone-implant integration.51
6.3 Mechanisms of Bioactive Material Function
Bioactive substances affect bone repair via a number of interrelated mechanisms:
- Formation of a hydroxycarbonate apatite (HCA) layer that is biologically active and creates a solid connection between the scaffold and native bone.
- Osteogenesis-related intracellular signaling pathways are triggered by ion release (Ca2+, Si2+, Mg2+, etc.).
- Improved cell adhesion and differentiation due to increased protein adsorption on the scaffold surface.
- Angiogenesis stimulation, which facilitates quicker waste elimination and nutrition flow at the regeneration site.52
5.4 Challenges and Future Outlook
Bioactive materials have showed a lot of promise, however problems including brittleness, unchecked deterioration, and low mechanical strength still exist. Furthermore, it is extremely challenging to achieve homogeneous dispersion of nanoparticles within polymer matrix.
Future research aims to:
- Developing multifunctional bioactive nano-scaffolds that can concurrently release growth factors and therapeutic ions is the goal of future research.
- Make customized, patient-specific scaffolds using sol–gel hybridization and 3D printing.
- Enhance surface modification to optimize immunological compatibility and bioactivity.
- Bioactive nano-scaffolds, which combine intelligent biological reactions with structural support, will serve as the foundation of next-generation regenerative implants with further innovation.55-60


 Bhumika Joshi*
Bhumika Joshi*
 Chanchal Jadhao
Chanchal Jadhao
 10.5281/zenodo.19464065
10.5281/zenodo.19464065