Heart failure with preserved ejection fraction (HFpEF) represents approximately half of all heart failure cases worldwide and continues to increase due to population aging and the rising prevalence of cardiometabolic disease. For many years, therapeutic options capable of improving outcomes in HFpEF were limited. Sodium?glucose cotransporter?2 (SGLT2) inhibitors have recently emerged as a major pharmacologic advance in the treatment of heart failure across the entire spectrum of ejection fraction. Large randomized trials have demonstrated that these agents reduce hospitalization for heart failure and improve clinical outcomes in patients with preserved ejection fraction regardless of diabetes status. This article reviews the mechanisms, clinical evidence, and practical implications of SGLT2 inhibitor therapy in HFpEF.
HFpEF, SGLT2 inhibitors, Heart failure, Cardiometabolic disease, Clinical outcomes
Heart failure with preserved ejection fraction is a complex clinical syndrome characterized by signs and symptoms of heart failure despite a left ventricular ejection fraction typically greater than or equal to fifty percent. Over the last two decades, HFpEF has become one of the most important challenges in cardiovascular medicine because its prevalence continues to increase worldwide. This rise is largely explained by population aging and by the growing burden of cardiometabolic conditions such as obesity, hypertension, diabetes mellitus, chronic kidney disease, and atrial fibrillation. In many regions, HFpEF now accounts for approximately half of all cases of heart failure, making it a major contributor to healthcare utilization, hospital admissions, and reduced quality of life (1,2).
The pathophysiology of HFpEF differs substantially from heart failure with reduced ejection fraction and involves multiple interacting mechanisms. Although systolic function is generally preserved, abnormalities in ventricular relaxation and compliance result in impaired diastolic filling and elevated left ventricular filling pressures. Structural and functional myocardial changes contribute to this process, including increased myocardial stiffness, interstitial fibrosis, and alterations in the extracellular matrix (3). In addition, endothelial dysfunction and microvascular abnormalities reduce nitric oxide bioavailability and impair myocardial perfusion, further contributing to myocardial stiffness and diastolic dysfunction. These processes are frequently amplified by systemic inflammation triggered by metabolic comorbidities such as obesity and diabetes, which promote vascular dysfunction and myocardial remodeling (4).
Patients with HFpEF commonly present with exertional dyspnea, reduced exercise tolerance, fatigue, and symptoms related to congestion such as peripheral edema or pulmonary congestion. These symptoms often fluctuate over time and are frequently associated with recurrent hospitalizations for acute decompensated heart failure. In contrast to heart failure with reduced ejection fraction, where several pharmacologic therapies have demonstrated clear mortality benefits, the management of HFpEF has historically been challenging. For many years, treatment strategies focused primarily on controlling comorbid conditions and relieving symptoms rather than modifying the progression of the disease (5,6).
Several pharmacological approaches have been evaluated in clinical trials, including renin-angiotensin system inhibitors, beta blockers, and mineralocorticoid receptor antagonists. Although these therapies provide benefits in specific clinical contexts, their overall impact on major outcomes in HFpEF has been modest or inconsistent (7). As a result, HFpEF has long been considered an area of significant unmet therapeutic need. The recent emergence of sodium-glucose cotransporter-2 inhibitors has therefore generated considerable interest, as growing evidence suggests that these agents may provide clinically meaningful benefits in patients with HFpEF regardless of diabetic status. The identification of an effective therapy for this complex syndrome represents a major step forward in the management of heart failure across the spectrum of ejection fraction (8,9).
DEVELOPMENT
SGLT2 inhibitors were initially developed for the treatment of type 2 diabetes mellitus as oral agents that lower plasma glucose by blocking glucose and sodium reabsorption in the proximal renal tubule. By inhibiting the SGLT2 transporter, these drugs increase urinary glucose excretion and induce a mild osmotic diuresis, which leads to small reductions in body weight and blood pressure in many patients. When the first large cardiovascular outcome trials in diabetes were conducted, investigators expected to confirm cardiovascular safety, but instead they observed an unexpected and consistent reduction in heart failure hospitalization. This finding was notable for two reasons. First, the separation of event curves in multiple studies appeared relatively early, suggesting that the benefit was not solely explained by slow changes in atherosclerosis or long-term glycemic control. Second, the magnitude of heart failure benefit seemed disproportionate to the modest changes in glucose levels, implying that SGLT2 inhibitors were modifying heart failure biology through mechanisms that were at least partly independent of diabetes (10). These observations changed the research trajectory of the drug class. What began as a glucose-lowering strategy rapidly evolved into a heart failure therapy program, leading to dedicated trials in patients with established heart failure and, ultimately, trials specifically addressing HFpEF and the spectrum of mildly reduced to preserved ejection fraction (11,12).
Several complementary mechanisms have been proposed to explain why SGLT2 inhibitors improve outcomes in HFpEF, a syndrome that is heavily influenced by congestion, cardiorenal interactions, and systemic cardiometabolic stress. A central and clinically intuitive mechanism is their effect on volume physiology. Through osmotic diuresis and natriuresis, SGLT2 inhibitors reduce circulating volume and lower ventricular filling pressures, which can translate into less pulmonary congestion and reduced symptoms such as exertional dyspnea. Importantly, many clinicians observe that the decongestive pattern of SGLT2 inhibitors appears “gentler” than aggressive loop diuresis, in the sense that it may preferentially reduce interstitial fluid while avoiding large swings in intravascular volume that can trigger hypotension or neurohormonal activation. In HFpEF, where congestion and elevated filling pressures are major drivers of hospitalization, even modest improvements in hemodynamic stability can have meaningful clinical impact (13).
Beyond volume reduction, SGLT2 inhibitors appear to influence the renal–cardiac axis, which is particularly relevant in HFpEF populations where chronic kidney disease and fluctuating renal function are common. HFpEF patients frequently cycle through episodes of congestion and diuretic escalation, followed by transient renal dysfunction, which then limits the clinician’s ability to optimize therapy and increases the risk of recurrent decompensation. SGLT2 inhibitors have been associated with kidney-protective effects in multiple clinical contexts, and in heart failure their renal profile is often characterized by a small early decrease in estimated glomerular filtration rate that stabilizes over time. Clinically, this matters because more stable kidney function can enable better long-term congestion control and reduce the cascade of events that leads to repeat hospital admissions. In practice, this is one reason SGLT2 inhibitors have been viewed as foundational rather than “optional,” particularly for patients with HFpEF who have diabetes, chronic kidney disease, or frequent congestion (14,15).
Metabolic and cellular mechanisms provide an additional layer of plausibility for benefit in HFpEF, especially given that many HFpEF phenotypes are closely linked to obesity, insulin resistance, and systemic inflammation. By creating a mild fasting-like metabolic state, SGLT2 inhibition can shift substrate utilization toward fatty acids and ketone bodies, which are considered efficient fuels for the myocardium under certain conditions. This shift may improve myocardial energetics and help the heart meet energy demands during stress or exercise, a period in which many HFpEF patients experience the greatest limitation. While the exact contribution of altered substrate metabolism to clinical outcomes remains an area of active investigation, it is consistent with the observation that benefits extend to patients without diabetes, and it aligns with the broader concept that HFpEF is not purely a hemodynamic disorder but also a cardiometabolic disease state (16).
SGLT2 inhibitors may also exert favorable effects on vascular function and inflammatory signaling, both of which are central themes in modern HFpEF pathophysiology. HFpEF is increasingly understood as a syndrome in which comorbidities drive systemic inflammation, endothelial dysfunction, microvascular impairment, and downstream myocardial stiffening. In experimental and translational work, SGLT2 inhibitors have been linked to reductions in oxidative stress and inflammatory mediators and to improvements in endothelial function, which could theoretically support better ventricular–vascular coupling and improved exercise capacity. These pathways are difficult to prove directly in outcome trials, but they offer a coherent explanation for why SGLT2 inhibitors might improve symptoms and quality of life, not just reduce hospitalization. In real-world care, even modest improvements in daily functioning can be highly meaningful for HFpEF patients who often live with chronic limitation, repeated acute episodes, and a heavy burden of comorbid disease (17,18).
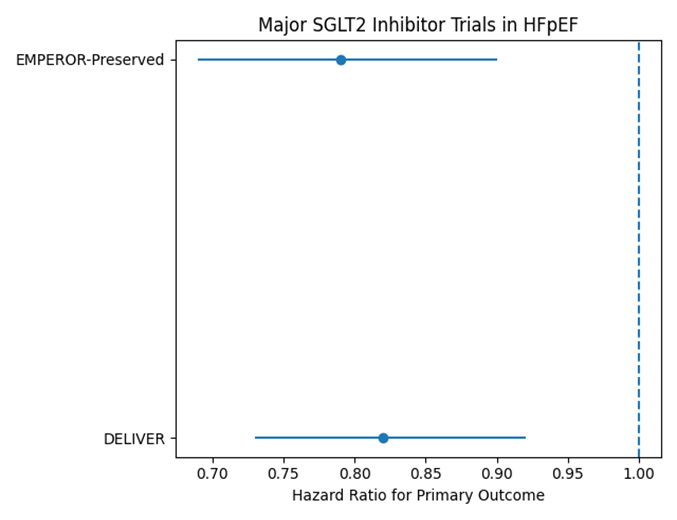
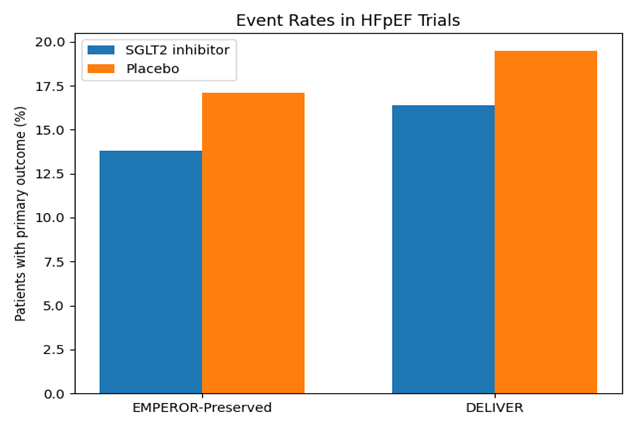
The clinical evidence supporting SGLT2 inhibitors in HFpEF is anchored by two landmark randomized trials that enrolled broad HFpEF and HFmrEF populations and tested fixed-dose therapy on top of usual care. The EMPEROR-Preserved trial evaluated empagliflozin in symptomatic patients with an ejection fraction greater than forty percent and demonstrated a significant reduction in the composite of cardiovascular death or hospitalization for heart failure, with a hazard ratio of 0.79. A critical point is that the observed benefit was largely driven by fewer heart failure hospitalizations, consistent with the concept that SGLT2 inhibitors stabilize congestion and reduce worsening heart failure events. The DELIVER trial evaluated dapagliflozin in a similar population across mildly reduced and preserved ejection fraction and also showed benefit on a composite endpoint of worsening heart failure or cardiovascular death, with a hazard ratio of 0.82. Across both trials, the direction of benefit was consistent in key subgroups, including patients without diabetes, reinforcing the idea that these drugs should be considered heart failure therapies rather than simply diabetes medications (19).
From a practical clinical standpoint, the trial evidence has several implications for routine HFpEF management. First, the benefits appear broadly applicable across typical HFpEF phenotypes encountered in practice, which supports early initiation once the diagnosis is established and the patient is clinically stable. Second, the simplicity of use is a major advantage: the trials used once-daily, fixed-dose regimens without complex titration, making implementation feasible in both cardiology and general internal medicine settings. Third, clinicians can frame expectations realistically: the most reliable outcome supported by the trials is a reduction in worsening heart failure events and hospitalizations, with additional patient-centered benefits suggested by improvements in symptoms and quality of life measures in heart failure research more broadly. Finally, the safety profile is generally manageable when clinicians apply standard precautions, particularly in patients prone to volume depletion, those on high-dose diuretics, or those with intercurrent illness that may increase risk of dehydration. This combination of mechanistic plausibility, robust randomized trial evidence, and pragmatic ease of implementation explains why SGLT2 inhibitors have rapidly moved into the core of HFpEF therapy and why they are now widely discussed as one of the first truly disease-modifying pharmacologic options for this historically difficult syndrome (18,15) .
DISCUSSION
The results of these trials have significantly changed the therapeutic landscape of HFpEF. For many years, the management of patients with preserved ejection fraction focused mainly on controlling comorbidities and relieving symptoms because pharmacological therapies failed to consistently improve major clinical outcomes. The demonstration that SGLT2 inhibitors reduce heart failure events represents a major shift in the treatment paradigm. The reduction in heart failure hospitalizations observed with SGLT2 inhibitors suggests that these agents improve hemodynamic stability and reduce congestion, which is a central pathophysiological component of HFpEF. By promoting natriuresis and osmotic diuresis, these drugs may decrease ventricular filling pressures and improve pulmonary congestion without causing excessive intravascular depletion, which is a frequent limitation of traditional diuretic therapy. As a result, patients may experience fewer episodes of acute decompensation requiring hospitalization, an outcome that has important implications for both patient prognosis and healthcare utilization (20).
In addition to reducing hospitalization rates, several studies have demonstrated improvements in patient-reported outcomes, including quality of life, functional status, and exercise capacity. These findings are particularly relevant in HFpEF, where symptom burden and reduced functional capacity significantly affect daily activities and overall well-being. Improvements in symptoms such as dyspnea and fatigue may translate into better tolerance for physical activity and improved participation in rehabilitation programs or daily activities. While mortality benefits in HFpEF remain less pronounced than in heart failure with reduced ejection fraction, the consistent reduction in worsening heart failure events and symptomatic improvement represents a meaningful clinical benefit for patients living with this chronic condition (21,22).
Another important aspect of SGLT2 inhibitor therapy is its favorable safety profile and ease of use in clinical practice. Unlike many traditional heart failure therapies that require careful dose titration and frequent monitoring, SGLT2 inhibitors are typically administered as fixed daily doses and can be initiated relatively easily in both outpatient and inpatient settings. This simplicity may facilitate broader implementation of therapy and improve adherence in real-world practice. The most commonly reported adverse effects include genital mycotic infections and mild volume depletion, both of which are generally manageable with appropriate patient education and monitoring. In most cases, these side effects do not require discontinuation of therapy. Rare adverse events such as diabetic ketoacidosis may occur in specific clinical contexts, particularly in patients with diabetes during acute illness or prolonged fasting, but overall the incidence remains low (20,23).
From a broader clinical perspective, the introduction of SGLT2 inhibitors has also influenced guideline recommendations and the conceptual framework of HFpEF management. Contemporary heart failure guidelines increasingly recognize these agents as foundational therapy across the spectrum of ejection fraction. Their benefits appear to be consistent regardless of diabetes status, renal function within trial inclusion ranges, or baseline ejection fraction within the preserved spectrum. This universality is particularly valuable in HFpEF, a syndrome characterized by considerable heterogeneity in clinical presentation and underlying pathophysiology. The ability of a single therapeutic class to provide benefit across diverse patient subgroups underscores its importance in contemporary heart failure care (24).
Another key point in interpreting these findings is the interaction between SGLT2 inhibitors and the cardiorenal axis. Many patients with HFpEF also suffer from chronic kidney disease, which complicates management and increases the risk of adverse outcomes. By stabilizing renal function and reducing congestion-related renal injury, SGLT2 inhibitors may help interrupt the vicious cycle in which heart failure exacerbations worsen kidney function and vice versa. This protective effect on renal physiology may contribute indirectly to improved cardiovascular outcomes and may also expand therapeutic options for patients who previously had limited treatment alternatives (23,24).
Despite these encouraging results, several questions remain regarding the optimal integration of SGLT2 inhibitors into the broader HFpEF treatment strategy. HFpEF is a heterogeneous syndrome with multiple phenotypes driven by distinct combinations of comorbidities, including obesity-related HFpEF, hypertensive heart disease, atrial fibrillation–associated HFpEF, and infiltrative cardiomyopathies. Future research will likely focus on identifying whether certain phenotypes derive greater benefit from SGLT2 inhibition and whether combination therapy with other emerging agents may further improve outcomes. Additionally, ongoing studies are exploring how SGLT2 inhibitors influence myocardial remodeling, inflammation, and metabolic pathways, which may provide deeper insight into their mechanisms of action (25).
Overall, the accumulating body of evidence suggests that SGLT2 inhibitors represent one of the most important therapeutic advances in HFpEF in recent decades. Their ability to reduce worsening heart failure events, improve symptoms, and provide renal protection, combined with a favorable safety profile and simple dosing strategy, makes them a valuable component of modern heart failure management. As clinical experience continues to grow and additional data emerge, SGLT2 inhibitors are likely to play an increasingly central role in the treatment of patients with HFpEF (25).


 Andrea Martinez Garay*
Andrea Martinez Garay*
 Julian Pereañez Martinez
Julian Pereañez Martinez
 10.5281/zenodo.19007689
10.5281/zenodo.19007689